Translate this page into:
Updates in molecules associated with pathogenesis of amoebiasis: A mini review
* Corresponding author: Dr. Rakesh Sehgal, Department of Microbiology, Aarupadai Veedu Medical College and Hospital, Vinayaka Mission’s Research Foundation-DU, Puducherry, India. sehgalpgi@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sharma D, Kaur U, Sehgal R. Updates in molecules associated with pathogenesis of amoebiasis: A mini review. South Asian J Health Sci. 2024;1:12–26. doi: 10.25259/SAJHS_11_2023
Abstract
Amoebiasis caused due to the protozoan parasite Entamoeba histolytica has been ranked as the third major reason of mortality after Malaria and Schistosomiasis. The pathogen adversely impacts the health of the children and hinders their development and can lead to intestinal and extra-intestinal manifestation, leading to more than 100,000 deaths annually. The most common route of transmission is via faecal oral route, but recent reports also suggest sexual transmission among homosexual men. Recent experiments in in-vitro, in-vivo and in-silico studies have contributed to a better understanding of pathogenesis by filling in the knowledge gaps related to adhesion, invasion, phagocytosis, encystation, trogocytosis and many other processes. Experimental studies evaluating the pathogenic potential of Entamoeba dispar also highlights the importance of considering other species under this genus. Despite the recent advances, there is a need for new drug targets due to the lack of a successful vaccine yet. This review therefore summarizes the recent molecules that have been identified and assessed to play a role in various pathogenic mechanisms of the parasite, further contributing to the progression of the disease.
Keywords
Entamoeba histolytica
pathogenesis
updates
new molecules
INTRODUCTION
Globally, 50 million people per year are infected with Entamoeba histolytica, the etiological agent of amoebiasis.[1] It is described as an enteric invasive protozoan pathogen. The World Health Organisation (WHO) has ranked amoebiasis as the third major reason of mortality because of parasitic ailments, accounting for over 100,000 deaths per year, especially in developing countries.[2] As it is prevalent in areas of poor sanitation, the cysts of Entamoeba are acquired easily by ingesting contaminated food and water.[3] The growth of children is also adversely affected due to E. histolytica-related diarrhoeal illnesses.[4] The National Institute for Allergy and Infectious Diseases (NIAID) classifies E. histolytica as a category B biodefense pathogen due to resistance to chlorine, easy spread through food and water, low infection doses, and environmental resistance.[5] In about 90% of cases, Entamoeba cysts asymptomatically colonise the intestinal lumen, leading to non-invasive disease and in another 10% population, trophozoites disrupt the mucosal barrier, invade the lamina propria and result in symptomatic infection.[6] The clinical manifestations vary from asymptomatic to severe signs, including extraintestinal abscesses and dysentery. Various organ systems might also be affected, such as respiratory, renal, genitourinary, and peritoneal locations, but among these, the most frequent manifestation is Amoebic Liver Abscess (ALA).[7]
The Entamoeba genus includes several species in which the intestinal lumen is the habitat of E. histolytica, E. dispar, E. moshkovskii, E. polecki, E. coli and E. hartmanni. The only species concerned with pathogenesis is Entamoeba histolytica, while others are considered non-pathogenic to humans.[8] Recent findings have demonstrated that patients with gastrointestinal symptoms are infected with E. dispar. But there is no conclusive proof between the existence of the organism and the host symptoms.[9] The pathogenic potential of E. dispar is still not clear. Hence, the objective of this review is to emphasise and offer a summary of the most recent molecules that have been identified to participate in the pathogenesis of E. histolytica. The progression of a disease, commonly referred to as pathogenesis, comprises five key phases: Exposure, adhesion, invasion, infection and transmission. The phases of exposure and transmission are most clearly understood when studying the parasite’s life cycle.
LIFE CYCLE
E. histolytica’s life cycle is described as monogenetic, as it requires only a single host, the humans. No secondary host is required, but some mammals have known to be with it infected by it.[10] Infection is caused by the intake of mature cysts, which are quadrinucleate [Figure 1]. These are mainly acquired through infected eatables, water or hands. The excystation takes place in the ileum and there is no effect of gastric juice on the chitinous cyst layer.[7] The cysts further transform into trophozoites by the action of trypsin, which disrupts the cyst wall. The quadric-nucleated amoeba emerges and proliferates further; eight amebula are then produced on division. They bind to the mucin layer of the intestine and continue to accumulate chromatid-like glycogen and black rod granules as their reserve food. It further forms a resistant cyst wall, completing the process of encystation, which occurs in the intestine lumen. The appearance of four nuclei marks the end of the development of cysts. An analysis on parasite Entamoeba invadens, infecting reptiles revealed that the encystation is initiated by the removal of galactose-binding lectin present on the surface membrane of pathogen.[11] On completion of the process, trophozoites present in loose stools (diarrhoea) and cyst in formed stools both are passed in the faeces. The cysts withstand the harsh environmental conditions for months due to their resistant walls. The trophozoites are destroyed in the gastric environment and also in the stool when they leave the body.[12] The asymptomatic colonisation can be described on the basis of the above, which accounts for 90% of infections. But the trophozoites colonise the intestine in the rest 10–20% of infections, which is done by the attachment of trophozoites to the large intestine’s mucin glycoprotein by a Gal/GalNAc-specific lectin.[12] These then obtain their nutrition and undergo binary fission. Neutrophils react to trophozoite intrusion and are accountable for colon disruption. Further invasion of the mucosal layer of the ileum by trophozoites gives them access to the bloodstream, which provides them a pathway to spread to the liver, lungs and other locations, resulting in various clinical forms. From these sites, the trophozoites or cysts cannot leave the host.[13]
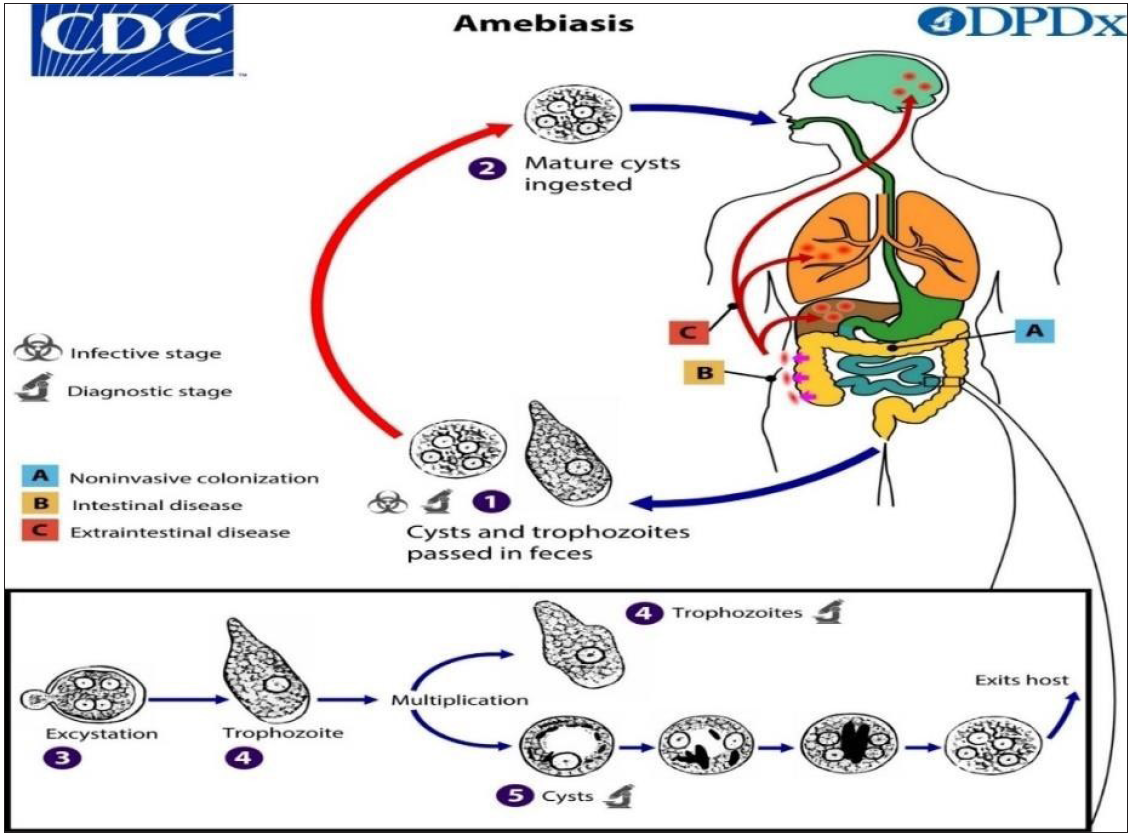
- Life cycle of Entamoeba histolytica (https://www.cdc.gov/parasites/amebiasis/pathogen.html)
Transmission: The primary routes of infection, despite the investments in better sanitation infrastructure, are the waterborne and foodborne transmission. Raw and undercooked food consumption increases the risk of foodborne transmission.[14] If food handlers do not ensure good sanitation, they can also lead to contamination. Flies and cockroaches further help in spreading the infection.[15] The use of biosolids as fertilisers often spreads bacteria through cross-contamination with animal waste, soiled implements, irrigation of polluted water, or sewage.[16] Oral–anal sexual practices among male homosexual men also lead to transmission.[17]
Reservoirs: Mostly humans and non-human primates are infected by Entamoeba. Till date, no well-defined zoonotic reservoir has been described except humans. The cysts on ingestion start dividing in the ileum, from where they reach the colon, and the epithelial lining is thereafter targeted by them.[15] The infection is further spread by cyst excreter. In other animals like cats, dogs, pigs, the infection is not frequent as they do not excrete cysts in their faeces. Furthermore, cysts are not able to survive outside the bodies. There were also no recorded vectors.[15]
Incubation period: It lasts from two weeks to three weeks, but this can further vary.[18] Around 4 to 10% of patients carrying E. histolytica experience clinical illness in 12 months. Even years after exposure, ALA can occur, which may further lead to the suppression of immune response.[6]
Communicability Time: People who are infected keep excreting the cysts in a recurrent way for years if they do not seek any treatment. The chances of transmission are poor in cases of acute amoebic dysentery since there are no cysts in dysenteric faeces and the fragile nature of trophozoites does not allow them to survive for a longer period of time. Also, after several years, patients with chronic infection will transfer the pathogen onto others via the excretion of cysts.[18]
RECENT ADVANCES IN THE PATHOGENESIS
Many studies involving various in vitro, in vivo methodologies, in silico investigations and docking analyses were conducted in the past year to analyse the functions of numerous molecules involved in the pathogenesis of Entamoeba histolytica. These have contributed to a better understanding of pathogenesis by elucidating various mechanisms that were previously unknown. The following sections highlight the newly identified compounds and their potential roles in Entamoeba histolytica pathogenesis adhesion, infection and invasion [Figures 2 and 3]. In the life cycle, the stages of exposure and transmission have already been defined.

- Molecules involved in different stages of (Made with MS powerpoint 2019).

- Diagrammatic representation of the main molecules involved (Made with MS powerpoint 2019).
ADHESION
Adhesion plays a key function in virulence and pathogenesis. It describes the motility, colonisation, nutrient uptake and tissue damage caused to the host. Rhomboid intramembrane proteases, like lectins, govern parasite–host interactions. Welter et al. investigated the link between rhomboid proteases and their effect on the lectin subunit submembrane position. They found that by lowering the activity of rhomboid protease EhROM1 using various ways, a shift in three Gal/GalNAc subunits was found on the cell membrane, including an escalation in the molecular weight of Hgl and Lgl, thereby shedding light on the molecular pathways that control parasite–host adhesion.[19] On the other hand, Rab GTPases are implicated in the cytolytic effect of E. histolytica as they interact with Gal/GalNAc lectins, which are further essential for the attachment to the intestinal cells as well as the production of virulence factors EhCP Cysteine proteinase. Batra et al. investigated EhRabX10, a pseudoRab GTPase, using physicochemical techniques and various in silico methods. They classified it as a pseudoRab GTPase having four non-conserved G-motifs, clearing the path for more research into the nucleotide identification, binding, and hydrolysis mechanisms.[20] Rab small GTPases also play a role in the cytoskeleton and signalling control. In the aetiology of E. histolytica, membrane trafficking, and cytoskeletal regulation are critical. The authors of another study characterised Rab7 in order to better understand its function. The cells in which EhRab7D was overexpressed, there was a reduction in erythrophagocytosis, trogocytosis of mammalian cells, further acidification and maturation of the phagosome. Transcriptional gene suppression of EhRab7D gene resulted in phagocytosis/trogocytosis and phagosome maturation phenotypes that were opposite. The findings show that EhRab7D has two opposing functions: a repressive role in phagocytosis/trogocytosis and lysosome/phagosome/trogosome formation, and a stimulatory involvement in adherence and motility, probably through interactions with specific effectors.
They also postulated a concept in which three EhRab7 isotypes were involved in phagocytosis/trogocytosis in a sequential manner.[21]
EhLMW-PTPs genes from E. histolytica encode for low molecular weight tyrosine. Phosphatases are found both in trophozoites and cysts but the role of these phosphatases in the parasite’s pathogenicity has yet to be determined.[22] Differential expression of the LMW-PTPs at the mRNA and protein levels in an asynchronous trophozoite culture was also evaluated further, discovering that trophozoites overexpressing EhLMW-PTP2 had lesser fibronectin adherence and a decreased cytopathic impact, possibly due to a reduction in phagocytic cups, exhibiting less erythrophagocytosis. These findings suggest that the parasite’s EhLMW-PTPs regulate the parasite’s proliferation, adhesion and phagocytosis mechanisms, which are all important in the parasite’s pathogenicity[22] [Table 1].
INVASION
When trophozoites encounter host cells, they may increase the expression of peroxiredoxins Prx as well as their secretion, implying that they have cell-to-cell contact-independent effects on macrophages. The C-terminal of peroxidoxin is accountable for triggering the NLRP3 inflammasome in macrophages, revealing parasites’ unique outside-in method for reducing NF-B-dependent pro-inflammatory responses by activating caspase-1 via NLRP3 and degrading cullin-1/5 from macrophages.[23] But during amoebic dysentery, trophozoites invade the colon wall and migrate through tissue layers that are rich in the extracellular matrix. The parasite’s capacity to move through tissue layers is a crucial need for invasion. Hasan et al. examined protein phosphorylation variations along with previously found hyperadherent E. histolytica cell line with impaired movement. They also discovered that EhCoactosin, an actin-binding protein that had never been recognised to be phosphorylated, is actually one of the differently phosphorylated proteins in E. histolytica. It is also responsible for the regulation of cell membrane dynamics and motility and together with other proteins, it was found to be differently phosphorylated, laying the framework for finding kinases and phosphatases which modulate tissue invasiveness.[24] Sharma et al., in their study, characterised E. histolytica type I PIPK EhPIPKI. The type I and III PIPK families have homologs, while type II PIPK was missing, according to the data obtained by computational analyses. Therefore, they concluded that the parasite contains a single EhPIPKI gene with unusual posttranslational processing features, including the existence of a domain that was repetitive and showed substrate selectivity that was absent from other PIPK enzymes[25] [Table 2].
| Molecule identified | Possible role in pathogenesis | Reference |
|---|---|---|
| Novel TLR4-Binding Domain of Peroxiredoxin | Activates the NLRP3 Inflammasome in macrophages | [23] |
| EhCoactosin | Coactosin phosphorylation controls the cell membrane dynamics and amoebic movement | [24] |
| EhPIPKI | Posttranslational processing and signalling mechanisms involved in invasion and pathogenesis | [25] |
INFECTION AND OTHER CELLULAR PROCESSES
Peroxisomes were earlier thought to be absent in E. histolytica, but in the research conducted by Verner et al. they provided the first evidence of peroxisomes being present in the parasite. They identified seven proteins accountable for peroxisome formation, which were Pex1,6,5,11,14,16, and 19 providing an insight into new possible mechanisms of pathogenesis.[26] The E. histolytica ESCRT-I complex is involved in vesicular trafficking and phagocytosis, and it also coordinates with other molecules that have a role in a similar process. Galindo et al. found that on knocking down the Ehvps23 gene it leads to a reduction in the frequency of phagocytosis and the vesicular structures which participate in vesicular trafficking. Antiviral defence, chromatin & transposon silencing, and other processes related to gene silencing are all controlled by RNA interference in parasites. There is not enough information about Entamoeba RNA-induced silencing complex RISC.[27] Zhang et al. studied the RISC of EhAgo2-2, Entamoeba’s highly expressed Argonaute protein, and revealed 43 protein constituents with a diverse range of functional activity. Two proteins with nucleosome assembly protein NAP domains were discovered to be novel core members of amoebic RISC, having never been detected before in other RNAi systems. This finding suggests that RNAi regulation may affect a variety of cellular functions in E. histolytica.[28] Protein phosphatase PP2C have been reported in E. histolytica, E. invadens and E. dispar. The molecule’s expression, activity and ultrastructural localisation changed between the cyst and trophozoite stages of E. invadens’ life cycle. Because the serine/threonine protein’s phosphorylation and dephosphorylation regulate different activities of host-parasite interactions, the Entamoeba PP2C found in this study might have a role in invasion, intracellular replication and the trophozoite encystation procedure.[29] Silencing of polyadenylation factor EhCFIm25 in E. histolytica altered trophozoite growth, mortality, and virulence, implying that EhCFIm25 could also be a new biochemical target.[30] Modulated proteins are mostly connected to glycolysis, carbon metabolism, cytoskeleton dynamics, parasite virulence, gene expression, and protein changes, according to the STRING analysis, functional GO-term annotations, and pathway analyses KEGG performed by the authors. More research is needed to corroborate the theories that have emerged from this proteomic study in order to provide a complete picture of the molecular pathways involved. Anaphase-promoting complex cyclosome is an E3 ubiquitin ligase that uses the ubiquitin-proteasome system to synchronise chromosomal segregation and anaphase advancement. EhApc11 from E. histolytica, a homologue of Apc11 in Homo sapiens, was chosen for thorough in silico research and molecular approaches to elucidate its structural and functional characteristics. EhApc11 is a powerful antigenic extracellular protein that is hydrophilic and thermostable. EhApc11 can have a possible relation with a variety of proteins involved in the cell cycle pathway, as well as with EhApc10. It’s crucial since human Apc10 has been linked to the regulation of the Apc2-Apc11 interaction.[31]
Endocytic mechanisms, such as phagocytosis, are critical for the protist parasite E. histolytica’s survival in the human gut. By combining a variety of actin-binding and regulating proteins, the actin cytoskeleton plays a part in the creation of pseudopods and phagosomal vesicles. Formin and profilin are two actin-binding proteins that are involved in modulating the endocytic processes by regulating actin cytoskeleton dynamics. Bharadwaj et al. found formin and profilin to be involved in phagocytosis and hypothesised that these two proteins interact and that the sequential recruitment at the site is needed for successful phagocytosis completion. Downregulation of formin expression results in the lack of profilin at the phagocytosis site, whereas profilin downregulation has no effect on formin localisation.[32] Pérez-Hernández et al. found that Gal lectin colocalises with c-Met in cocultures of E. histolytica trophozoites and HepG2, indicating that the two molecules interact and that contact with c-Met is required for cytotoxicity. TOR kinases regulate cell growth and proliferation through a variety of signalling pathways. EhFRAP and EhTOR2 are two genes in E. histolytica that code for TOR-like proteins. Structural resemblances are found with TOR kinases in case of TOR amoebic homologs. Both of them have the ability to form TORC complexes and take part in protein–protein interaction networks that contribute to pathogenesis, according to the findings.[33] The structure–function connections of EhFRAP and EhTOR have never been characterised in silico before.[34]
NETosis is a neutrophil process that includes a series of processes from pathogen recognition through the release of DNA containing antimicrobial proteins, as well as the primary creation of NADPH oxidase dependent or independent reactive oxygen species. Earlier, the authors observed that NETosis activated by E. histolytica trophozoites is independent of neutrophil NADPH oxidase activity but is dependent on parasite survival, with no ROS source detected. These findings imply that E. histolytica-induced NETosis requires ROS produced by trophozoites and processed by extracellular MPO during interaction with neutrophils.[35] SUMOylation has yet to be investigated in E. histolytica. Its importance in phagocytosis, along with the role played by E. histolytica SUMO gene and its product were studied.[36] It was found to have an elongated N-terminus, which distinguishes it from SUMO and ubiquitin, according to the findings. The GG residues at the C-terminus and the KXE/D binding motif, both of which have a role to play in target protein interaction, were also shown. Phagocytic channels had sumoylated proteins present around the swallowed RBC’s. The association of EhSUMO with EhADH was predicted by docking analysis. The immunoprecipitation and immunofluorescence techniques showed that it was higher during phagocytosis; however, in the EhVps32, which is an ESCRT-III complex protein, the EhSUMO association seemed stronger under basal conditions. Lipid transfer proteins are essential for distinct organelle distribution of lipids and cellular lipid homeostasis.[36] Das et al. discovered a new role for LTPs in parasitic protist E. histolytica phagocytosis, trogocytosis, pinocytosis, biosynthetic secretion, pinosome recycling and motility. They demonstrated that two LTPs with StAR-related lipid transfer START domains EhLTP1 and EhLTP3 are active in various biological processes in an LTP-specific manner. The discoveries deliver new proposals for LTPs that are important for understanding the pathophysiology of parasitic protist-caused illnesses.[37]
Protein arginine methylation controls a variety of cellular processes like epigenetics, splicing, translation and stress response. Protein arginine methyltransferases PRMTs catalyse this posttranslational alteration, which are categorised into four types based on the molecules they produce. E. histolytica possesses four PRMTs related to type I, out of which three are comparable to PRMT1, but the fourth doesn’t display enough homology to be placed in any known PRMT family, hence it is referred to as an atypical PRMT EhPRMTA. The native enzyme changed its expression level after heat shock and erythrophagocytosis, and the recombinant EhPRMTA showed catalytic activity on commercial histones in an experimental set-up. Furthermore, knocking down EhPRMTA increased cell development and phagocytosis but decreased the cell migration and survival of trophozoites exposed to heat shock, implying the role of protein in regulating these activities. As a result, the findings imply that this methyltransferase is involved in cellular activities associated with virulence and cell survival [38]. The presence of GATA transcription factor in E. histolytica and its role as regulator of phagocytosis-related genes was investigated further.[27] In those trophozoites where EhGATA was expressed more, the amount of Ehadh and Ehvps32 mRNAs was altered, indicating the role of EhGATA in their transcription. The morphology was also altered with trophozoites depicting more spherical shape, an increase in the level of proliferation, and a decrease in the level of erythrophagocytosis. These findings therefore, shed light on EhGATA’s activity as a non-canonical transcription factor that regulates the genes involved in phagocytosis.[27]
Peroxiredoxin, a key antioxidant enzyme for parasite survival during the invasion phase, was found to trigger autophagy in macrophages in a study.[23] In order to establish infection within the host, pathogens must hijack the autophagy system, which is a crucial element of innate immunity. In RAW264.7 cells and mice, autophagosomes were observed in macrophages treated with recombinant Prx for 24 hours. Immunofluorescence and immunoblotting techniques were used to detect this. RAW264. Seven macrophages developed cytotoxicity due to Prx when the time period was increased to 48 hours, which was attributable to autophagy-dependent cell death. According to RNA interference studies, Prx stimulated autophagy via the TRIF pathway TLR4-TIR domain containing adaptor-inducing interferon. The key functional area for autophagy activation was the 100-amino-acid C-terminal part of Prx. These data points to E. histolytica Prx as a possible cause of autophagy and cytotoxicity in macrophages, indicating a potential pathogenic mechanism for a parasite.[23] E. histolytica needs a lot of iron to survive, therefore, when it invades, it utilises iron proteins from the host. Iron is delivered to all the cells with the help of transferrin, which is a plasma iron-binding protein. By attaching to an amoebic TfR EhTfR, iron-loaded Tf holoTf in humans can stimulate amoebae proliferation in vitro, and amoebae endocytose it inside clathrin-coated vesicles. In this study, it was discovered that EhTfR phosphorylation is essential for E. histolytica to endocytose human holoTf. Human holoTf could be destroyed by cysteine proteases for nutritional purposes once this complex is endocytosed. With the involvement of phosphoinositide 3-kinase PI-3 K and Ca2+, holoTf endocytosis activates the mitogen-activated protein kinases MAPKs and focal adhesion kinase FAK pathways, which stimulate cell proliferation. The study of these proteins and their signalling pathways could aid in the development of future treatments.[39] In several eukaryotic pathogens, second messenger signalling regulates a remarkable variety of activities. The cAMP signal transduction pathway of E. histolytica was studied using the existing genetic data available. In its genome, it has three heptahelical proteins with varying G-protein-coupled receptor domains, heterotrimeric G-proteins, soluble adenylyl cyclase, cyclase-associated protein and the enzyme carbonic anhydrase. They noticed variations in intracellular cAMP levels as well as a deficiency in the parasite E. histolytica’s rate of phagocytosis of red blood cells when they used particular inhibitors against key identified targets. The scientists believe that characterising some of these previously unknown critical signalling determinants will lead to a paradigm shift in our knowledge of this organism’s pathogenicity.[40]
Extracellular vesicles, EVs, which are released by cells to move biomolecules to the outside environment, are vital in cell–cell interaction via cargo transfer. Sharma et al. recognised and characterised these vesicles from Entamoeba. Exosome marker proteins, such as proteins involved in the development of these vesicles, cell signalling, metabolism and cytoskeletal proteins, were prominent in EV proteome, as analysed via mass spectrometry. EVs were also discovered to selectively package small RNAs, which comprised a subset of the cellular antisense small RNA population previously identified in Entamoeba RNA interference RNAi pathway proteins, including Argonaute. Surprisingly, they discovered that amoebic EVs affected parasite intercellular communication and encystation efficiency. EVs from encysting parasites aided encystation in other parasites, whereas EVs from metabolically active trophozoites hampered it. These findings show that parasite releases these vesicles that are alike in both the size and structure of exosomes identified earlier from other parasites and also, EVs have a specified protein and short RNA payload, playing a role in parasite intercellular communication and growth dynamics.[41]
Pinotsis et al. undertook a study to describe the control of E. histolytica actinin 2 EhActn2 by Ca2+Calcium. Ca2+ free and Ca2+ bound EhActn2 crystal structures revealed a calmodulin-like domain, which was discovered to be located within the rod domain. Various integrative investigations have demonstrated that the EhActn 2 calmodulin has a high affinity for calcium ions. The concentration of magnesium ions further plays a role in this. Calcium ion binding increases the rigidity of multidomain proteins; after that, interdomain cross-talk limits the conformational flexibility of F-actin-binding domains, impeding F-actin bundling. In vivo research conducted by the same group revealed that EhActn2 is involved in the development of phagocytic cups.[42] Transporting receptors control the appropriate targeting and release of lysosomal hydrolases. CPBFs are also present in E. histolytica which attach to a wide variety of cargos, like cysteine proteases and glycosidases, which are involved in disease progression. Research on CPBF2, a protein implicated in cell motility and extracellular matrix invasion, was carried out. Surprisingly, these functions of CPBF were unrelated to its cargo that is alpha-amylase. This is the first proof that a putative hydrolase receptor is involved in parasitic protozoa cell motility and invasion.[43]
Various biological functions like cell polarity, gene transcription, microtubule dynamics and cell cycle are regulated by GTPases that function as molecular switches. Verma et al. identified an E. histolytica Ca2+-binding protein EhCaBP6 as a new GTPase. They discovered the active location for GTP hydrolysis within EhCaBP6’s C-terminal domain, and also found that GTP binding causes EhCaBP6 to change conformation. The discovery of this novel and atypical Ca2+-dependent GTPase is critical for understanding E. histolytica’s peculiar cell cycle. Rab21 is a GTPase that has been discovered in a wide range of eukaryotic cells.[44]
The findings of a study conducted by Constantino-Jonapa et al. suggest that Rab 21 is involved in maintaining the structure of the Golgi apparatus, a vesicular trafficking organelle. Furthermore, proteins like Rab1A and Sec24, which were found to be connected to EhRab21 in this work, have a twofold function: first, they maintain the organelle structure, and second, they transport COPII vesicles from the endoplasmic reticulum to the Golgi apparatus. Lysosome development was also influenced by EhRab21. Using hamsters Mesocricetus auratus as an ALA formation model, the role of EhRab21 in the pathogenesis of amoebiasis was confirmed. The overexpression of EhRab21Q64L, a type of positive dominant mutant protein, resulted in a reduction in the number of liver abscesses.[45] Phosphatidylinositol phosphates PIPs play a vital role in a variety of cellular processes. PI3-phosphate, PI3P, is important for phagosome/trogosome biogenesis because it recruits PI3P effectors. Watanabe et al. used a novel technique to discover the proteins PI3P, which were dependently recruited to phagosomes because no known PI3P downstream effectors are preserved in E. histolytica. EhSNX1 and EhSNX2, the two putative SNXs, were discovered in the genome. EhSNX2 was recruited to trogosomes while EhSNX1 was attracted to the trogocytic cup and tunnel-like shaped structures, as seen via live and immunofluorescence imaging. Silencing the EhSNX2 gene increased trogocytosis, implying that it is a trogocytosis inhibitor. Many cellular functions in the amoebic cell need the remodelling of the actin cytoskeleton in response to external inputs. EhC2B, a C2 domain-containing protein, was shown to be an actin regulator in this study.[46] The findings of Tripathi et al. suggested that EhC2B works as a molecular bridge that promotes membrane deformation in a calcium-dependent way throughout the evolution of the phagocytic cup via its actin nucleation activity. Entamoeba’s virulence and survival are linked to the parasite’s motility and phagocytosis. These are regulated through actin-dependent mechanisms.[47] During phagocytosis, the scientists looked at the involvement of PtdIns4,5P2 and the enzyme that produces EhPIPKI. EhPIPKI was found to be enriched at various phases of phagocytosis, from onset to cup closure, in immunofluorescence and live cell pictures. However, after phagosomes were squeezed away from the membrane, the enzyme was no longer present. The development of phagocytic cups was reduced, and the frequency of erythrocyte engulfment was inhibited when a dominant negative mutant was upregulated. Furthermore, with the exception of PIPKs from other animals, EhPIPKI specifically binds to F and G-actin. A GFP-tagged PH-domain from PLC, which exclusively binds PtdIns4,5P2 in trophozoites, indicated that the enzyme’s product, PtdIns4,5P2, follows the same distribution pattern during phagocytosis. In conclusion, EhPIPKI controls actin dynamics to regulate phagocytosis initiation.[25]
Mobility and phagocytosis are critical elements in E. histolytica’s colonisation and infiltration of tissues. The functional molecules required during phagocytosis are transported to their correct cellular location with the help of EhRab proteins. During phagocytosis, the interaction of EhRabB with EhCPADH and actin was investigated. By preventing actin polymerisation, which was achieved by Latrunculin A, the scientists evaluated the role of the actin cytoskeleton. The mobility of EhRabB and EhCPADH, and the rate of phagocytosis were impacted. The association of this molecule with EhCPADH was demonstrated in mutant trophozoites with the EhRabB gene being silenced, reinforcing the involvement of actin during erythrophagocytosis.[48] Gronebery et al. found that via IL-6, HIF-1a modifies the immune response, resulting in different disease susceptibilities in males and females.[49] While fighting off the host’s defences, E. histolytica damages its plasma membrane, PM, resulting in viability loss. However, it is unknown if the trophozoites are able to repair the harm. The following research looked at six genes that code for aSMaes and were transcribed in the HM1:IMSS strain. In basal growth circumstances, the EhaSM6 gene was shown to be the most transcribed and also created a functional protein. The findings reveal that the aSMase6 enzyme aids in the repair of pore-forming molecules and damaged plasma membranes, thereby preventing the loss of cell integrity. Therefore, when confronted with the host’s lytic defence systems, this unique system may hence act further [Table 3].[50]
| Molecule identified | Possible role in pathogenesis | Reference |
|---|---|---|
| Peroxins Pex1, 6, 5, 11, 14, 16 & 19 | Proteins involved in the formation of peroxisomes | [26] |
| EhVps23 | ESCRT-I component involved in vesicular trafficking and phagocytosis | [51] |
| RISC of EhAgo2-2 | There were 43 protein hits identified with activities ranging from transcription, translation to sRNA loading and other cellular functions | [28] |
| Protein Phosphatase PP2C | Possible role in invasion, intracellular replication and encystation | [29] |
| EhCFIm25 | Trophozoite proliferation, death and pathogenicity | [30] |
| EhApc11 | Interacts with other proteins EhApc10 which are involved in cell cycle pathway | [31] |
| Amoebic formin and profilin | Phagocytosis | [32] |
| EhFRAP and EhTOR2 | Targets for targeted inhibition in E. histolytica, resulting in the disruption of TOR-associated signalling cascades | [34] |
| EhSUMO | Phagocytosis | [36] |
| EhLTP1 and EhLTP 3 | Endocytosis, exocytosis and motility | [37] |
| EhPRMTA | Proliferation of cells, heat shock response and regulation of virulence in vitro | [38] |
| EhGATA | Regulation of genes associated with phagocytosis | [27] |
| Peroxiredoxin | Activates autophagy and cytotoxic effects in macrophages | [52] |
| EhActn2 | Phagocytic cup formation | [42] |
| CPBF2 | Motility and invasion of the parasite | [43] |
| EhCaBP6 | Cell proliferation | [53,44] |
| EhC2B | Phagocytosis of RBCs through actin nucleation | [47] |
| EhRab 21 | Actin cytoskeleton regulation, lysosomal biogenesis and Golgi structuration | [45] |
| EhSNX1 and EhSNX2 | Trogocytosis | [46] |
| EhPIPKI | By modulating actin dynamics, it controls the initiation of phagocytosis | [25] |
| EhRabB | During phagocytosis, it mobilises the EhCPADH complex via the actin cytoskeleton | [48] |
| EhaSM6 | To avoid losing cell integrity, supports the repair of PMs that have been damaged by pore-forming chemicals | [50] |
| EhRab7D | Phagocytosis, trogocytosis and cytoskeletal reorganisation | [21] |
| Eh RabX10, a pseudoRab GTPase | Participation in the Rab-Sec signalling cascade involved in vesicular pathway in later stages | [20] |
TRANSMISSION
Cysts are the only way for amoebiasis to spread. Lipids are most likely among the metabolites that play a role in encystation. However, there hasn’t been a full lipid analysis published because the majority of the important lipid metabolic pathways are unknown. By using lipidomics, Mi- ichi et al. discovered dihydroceramides Cer-NDSs with very long N-acyl chains, C26 to C30, as critical metabolites for encystation. They also discovered that these Cer-NDSs are required for the parasite to display dormancy as it repels substances and avoids water loss by generating membrane impermeability. As a result, Entamoeba’s parasitic existence depends on ceramide metabolism. During cyst formation, the parasites create a wall structure surrounding it to confer resistance. Chitin along with other proteins, like chitinase, makes up the cyst wall. To better understand the role of chitinase in cyst development, the authors also screened a library of 400 chemicals for a new chitinase inhibitor.[54,55] They demonstrated that inhibiting chitinase induces the creation of aberrant cyst walls, with the formation of ‘pot-like structure’, using the inhibitor. As a result, the number of mature cysts produced decreased. Entamoeba must consequently create mature cysts in order to transmit to a new host, which necessitates the use of chitinase. Cysts survive in harsh environments and are further spread by contaminated food and water, being responsible for disease transmission from one host to another.[54 55] Manna et al. found a novel transcription factor called Encystation Regulatory Motif-Binding Protein, or ERM-BP. It plays an important part in encystation and responds to NAD+. When a mutation in the ERM-BP-DBM DNA binding domain and the ERM-BP-C198A Nicotinamidase domain resulted in protein mislocalisation, the efficiency of encystation was lowered. The importance of ERM-BP in stress response was also underlined in this study. The NAD+ responsive transcription factor is implicated in both development and the response to heat shock [56] [Table 4].
| Molecule identified | Possible role in pathogenesis | Reference |
|---|---|---|
| Chitinase | Formation of a mature round cyst | [54] |
| Dihydroceramides Cer-NDSs | By switching membrane characteristics, the Cer-NDS lipid subclass plays a critical role in Entamoeba cell development and morphogenesis | [55] |
| Encystation Regulatory Motif-Binding Protein ERM-BP | Encystation, its role in development, and how it responds to heat shock | [56] |
IMMUNOPATHOGENESIS
The infection associated with E. histolytica induces an immune response in the host’s body. Reactive oxygen species are known to play both protective as well as destructive role in E. histolytica. Therefore, the parasite has oxidative stress defense mechanisms due to the excessive ROS production. L-cysteine, flavodiiron proteins, peroxiredoxin and trichostatin A are the antioxidants that contribute to both the virulence as well as the defense mechanism. Cell death also occurs by the processes of apoptosis and necrosis, resulting in damage to the host.[35]
Begum et al. showed that inducing the production of an alarmin molecule, HMGB1, during Entamoeba infection has a dual purpose. It functions as both a chemoattractant and a pro-inflammatory cytokine. In the parasite, this chemical plays a critical role in shaping the pro-inflammatory environment. The mechanism by which E. histolytica binds to macrophages at the intercellular interface in disease pathogenesis and produces severe pro-inflammatory responses is unknown. The parasite overtakes the autophagy mechanism in macrophages to cause a malfunctioning in the pro-inflammatory responses. On the direct interaction of live trophozoites with macrophages, it triggered caspase-6, which resulted in the fast proteolytic disintegration of the protein complex associated with autophagy ATG16L1 as it wasn’t dependent on the stimulation of NLRP3 inflammasome, along with caspase-3/8, implying that the ATG16L1 protein complex has a role to monitor the modifying pro- inflammatory response in amoebiasis. It has been hypothesised that androgens are a factor behind the increased risk of developing monocyte-mediated pathologies.[57] Supporting this hypothesis Sellau et al. found that in castrated males which were suffering from hepatic amoebiasis, an elevation in the frequency of TNF/CXCL-1 producing classical monocytes was observed. Due to its similarities to trogocytosis in other organisms, Entamoeba is known to kill host cells in a similar manner.[58] Miller et al. showed that E. histolytica obtains and presents human cell membrane proteins via trogocytosis, resulting in protection from human serum lysis. The findings are the initial ones to show the display of human cell membrane proteins by amoebae, implying that membrane protein attainment and presentation are a common aspect of trogocytosis. These findings have substantial ramifications for E. histolytica’s interactions with the immune system, as well as also revealing a unique pathogen immune evasion technique[59] [Table 5].
Co-infection
Coinfection with virulent organisms, particularly enteropathogenic bacteria, is one of the reasons that have been suggested to contribute to the change of the usual intestinal microbiota, immunological modulation and, as a result, enhancing the virulence of the parasite. The interactions of Entamoeba with enteropathogenic Escherichia coli bring about a change in the virulence factors, thereby modifying the innate immune responses of the host which are linked to disease progression.[61]
ASYMPTOMATIC INFECTION
Yanagawa et al. in their study provided an insight into the pathophysiology of asymptomatic infections where the infection sites were restricted to the proximal colon where trophozoites were present, but the cystic forms were frequently found in faeces. Asymptomatic invasive cases had a more homogeneous gut microbiome composition than symptomatic invasive cases, with a higher abundance of Ruminococcaceae, Coriobacteriaceae and Clostridiaceae and a lower abundance of Streptococcaceae.[62]
Entamoeba dispar
It infects about 12% of the global population and was formerly categorised as non-invasive.[2] In a study by da Silva et al. ACFN strain was found to be the most pathogenic. The second pathogenic strain was MCR. The findings showed that E. dispar trophozoites possess the capability of phagoerythrocytosis, but in lesser proportions than E. histolytica. Pathophysiology was characterised by large areas of necrosis and well-developed chronic granulomatous inflammation.[63] KERP1 has been recognised as a virulence factor, which is expressed on the cell surface. It is a protein rich in glutamic acid and lysine. KERP1 was earlier not assumed to be present in E. dispar but Weedall et al., 2020 in their study found the gene to be present in E. dispar and E. moshkovskii. Infection with E. histolytica is generally linked with neutrophil infiltration.[64] The presence of trophozoites activates the neutrophil extracellular traps. However, there is no evidence that non-pathogenic E. dispar can cause NET formation. While looking into the potential of E. dispar to cause NET formation or not, E. histolytica-induced NET was significant for capturing and killing amoebas; E. dispar, on the other hand, did not cause NET formation, neutrophil degranulation or the formation of reactive oxygen species under any circumstances. These findings demonstrate that only live pathogenic E. histolytica stimulates neutrophils to make NET, and that the parasite is recognised by a carbohydrate with an axial HO- group at the fourth carbon.[65]
COVID-19 AND AMOEBIASIS
Although no studies have looked at the part played by viruses in promoting E. histolytica infection, SARS- CoV2 infection is identified as a factor that can cause the intestinal homeostasis to be dysregulated. Severe inflammation is the outcome of this invasion/infection as immune cells respond to it. In case of COVID-19-infected patients, intestinal bacterial diversity is known to decline. The microbes opportunistic in nature, like Streptococcus, Rothia, Veillonella, and certain fungi like Aspergillus and Candida spp. depict an increase in their relative abundance, whereas that of useful symbionts is reduced.[66,67] Invading viruses can both govern and regulate the ecosystem of commensal bacteria, facilitating either stimulatory or suppressive actions. Corticosteroids, which are commonly used to treat severe COVID-19, may have a part in this process by altering gut flora and favouring parasite infection. Although the mechanisms by which asymptomatic infection and dysenteric colitis progress into an ALA are unknown, these undiscovered pathways could provide an insight into anti-amoebic and anti-SARS-CoV-2 responses. SARS-CoV2 may influence the cytokine storm. The consequences of this are inflammation along with tissue destruction. Firstly, it compromises the effectiveness of the serum complement system, secondly, the secretion of IFN-y is inhibited, and lastly, the composition of gut microbiome is also altered. These factors may, therefore, contribute to or enhance the development of the extraintestinal form.[66,67] COVID-19 can exacerbate parasitic infections, which are already present, but recent findings suggest that it may also serve a preventive effect, mediated via the generation of Interferon- gamma.[68]
CONCLUSION
The socioeconomic settings in which E. histolytica thrive remains a big problem in developing countries. Although awareness regarding hygiene practices has resulted in people paying a certain degree of attention, there is still a long way to go. Vaccines have yet to be developed. Furthermore, resistance to currently used medications has been discovered, underlining the need for novel treatment techniques to be developed, and recent discoveries of novel pathogenic mechanisms have opened the road for it. Understanding the biology of the parasite will need the identification of new molecules and their likely roles in various mechanisms such as invasion, adhesion and infection. These new results may be useful in deciphering the pathophysiology of various other protist infections. E.dispar’s pathogenic potential has also been underlined in new research. Therefore, further studies are required for a deeper insight into its pathology. These breakthroughs also establish the groundwork for further research to be done in the future.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study
Financial support and sponsorship
Nil.
Conflicts of interest
Dr. Rakesh Sehgal is on the Editorial Board of the Journal
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Intestinal amoebiasis: 160 years of its first detection and still remains as a health problem in developing countries. International Journal of Medical Microbiology. 2020;3101:151358. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/S1438422119303467
- [CrossRef] [PubMed] [Google Scholar]
- WHO/PAHO/UNESCO report. A consultation with experts on amoebiasis. Mexico City, Mexico 28-29 January, 1997. Epidemiol Bull. 1997;18:13-4.
- [PubMed] [Google Scholar]
- Tissue destruction and invasion by Entamoeba histolytica. Trends in parasitology. 2011;276:254-63. [accessed 2023 Nov 9] Available from: https://www.cell.com/trends/parasitology/fulltext/S1471-49221100039-0
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Entamoeba histolytica-associated diarrheal illness is negatively associated with the growth of preschool children: evidence from a prospective study. Transactions of the Royal society of tropical Medicine and Hygiene. 2006;10011:1032-8. [accessed 2023 Nov 9] Available from: https://academic.oup.com/trstmh/article-abstract/100/11/1032/1940784
- [CrossRef] [PubMed] [Google Scholar]
- A review of the global burden, new diagnostics, and current therapeutics for amebiasis. Open forum infectious diseases 2018:ofy161. [accessed 2023 Nov 9] Available from: https://academic.oup.com/ofid/article-abstract/5/7/ofy161/5049601
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Amoebiasis. The lancet. 2003;3619362:1025-34. [accessed 2023 Nov 9] Available from: https://www.thelancet.com/article/S0140-67360312830-9/abstract
- [CrossRef] [PubMed] [Google Scholar]
- Amebiasis. New England journal of medicine. 2003;34816:1565-73. [accessed 2023 Nov 9] Available from: https://www.nejm.org/doi/full/10.1056/nejmra022710
- [CrossRef] [PubMed] [Google Scholar]
- Ribosomal RNA genes of ‘pathogenic’and ‘nonpathogenic’Entamoeba histolytica are distinct. Molecular and Biochemical Parasitology. 1991;492:297-302. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/016668519190073F
- [CrossRef] [PubMed] [Google Scholar]
- Entamoeba moshkovskii and Entamoeba dispar-associated infections in Pondicherry, India. Journal of Health, Population and Nutrition 2005:292-5. [accessed 2023 Nov 9] Available from: https://www.jstor.org/stable/23499331
- [PubMed] [Google Scholar]
- Differentiation of Entamoeba histolytica and Entamoeba dispar Cysts Using Polymerase Chain Reaction on DNA Isolated from Faeces with Spin Columns. European Journal of Clinical Microbiology & Infectious Diseases. 2000;195:358-61. Jun 5 [accessed 2023 Nov 9] Available from: http://link.springer.com/10.1007/s100960050494
- [PubMed] [Google Scholar]
- Crithidia fasciculata induces encystation of Entamoeba invadens in a galactose-dependent manner. The Journal of parasitology 1998:705-10. [accessed 2023 Nov 9] Available from: https://www.jstor.org/stable/3284574
- [PubMed] [Google Scholar]
- A Role for a Galactose Lectin and Its Ligands During Encystment of Entamoeba 1. J Eukaryotic Microbiology. 2001;481:17-21. Jan [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1111/j.1550-7408.2001.tb00411.x
- [CrossRef] [PubMed] [Google Scholar]
- Intestinal invasion by Entamoeba histolytica shahram solaymani-mohammadi. Sub-cellular biochemistry. 2008;47:221. [accessed 2023 Nov 9] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2649828/
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Risk factors for infection by the Entamoeba histolytica/E. dispar complex: an epidemiological study conducted in outpatient clinics in the city of Manaus, Amazon Region, Brazil. Transactions of the Royal society of tropical Medicine and Hygiene. 2005;997:532-40. [accessed 2023 Nov 9] Available from: https://academic.oup.com/trstmh/article-abstract/99/7/532/1920644
- [CrossRef] [PubMed] [Google Scholar]
- Opportunistic amoebae: challenges in prophylaxis and treatment. Drug Resistance Updates. 2004;71:41-51. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/S1368764604000056
- [CrossRef] [PubMed] [Google Scholar]
- Tissue invasion by Entamoeba histolytica: evidence of genetic selection and/or DNA reorganization events in organ tropism. PLoS neglected tropical diseases. 2008;24:e219. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0000219
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Amebiasis in HIV-1-infected Japanese men: clinical features and response to therapy. PLoS neglected tropical diseases. 2011;59:e1318. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0001318
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Control of Communicable Diseases Manual. American Public Health Association. 2008.
- Reduced expression of a rhomboid protease, EhROM1, correlates with changes in the submembrane distribution and size of the Gal/GalNAc lectin subunits in the human protozoan parasite, Entamoeba histolytica. PloS one. 2020;153:e0219870. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0219870
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Exploring insights of syntaxin superfamily proteins from Entamoeba histolytica : a prospective simulation, protein‐protein interaction, and docking study. J of Molecular Recognition. 2021;346:e2886. Jun [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1002/jmr.2886
- [CrossRef] [PubMed] [Google Scholar]
- Rab7D small GTPase is involved in phago‐, trogocytosis and cytoskeletal reorganization in the enteric protozoan Entamoeba histolytica. Cellular Microbiology. 2021;231 Jan [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1111/cmi.13267
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Low molecular weight protein tyrosine phosphatase LMW-PTP2 protein can potentially modulate virulence of the parasite Entamoeba histolytica. Mol Biochem Parasitol. 2021 Mar;242:111360.
- [CrossRef] [PubMed] [Google Scholar]
- A Novel TLR4-Binding Domain of Peroxiredoxin From Entamoeba histolytica Triggers NLRP3 Inflammasome Activation in Macrophages. Front Immunol. 2021;12:758451.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Coactosin Phosphorylation Controls Entamoeba histolytica Cell Membrane Protrusions and Cell Motility. Johnson PJ, editor. mBio. 2020;114:e00660-20. Aug 25 [accessed 2023 Nov 9] Available from: https://journals.asm.org/doi/10.1128/mBio.00660-20
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Novel regulatory roles of PtdIns4,5P 2 generating enzyme EhPIPKI in actin dynamics and phagocytosis of Entamoeba histolytica. Cellular Microbiology. 2019;2110 Oct [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1111/cmi.13087
- [CrossRef] [PubMed] [Google Scholar]
- Anaerobic peroxisomes in Entamoeba histolytica metabolize myo-inositol. PLoS pathogens. 2021;1711:e1010041. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1010041
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A noncanonical GATA transcription factor of Entamoeba histolytica modulates genes involved in phagocytosis. Molecular Microbiology. 2020;1146:1019-37. Dec [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1111/mmi.14592
- [CrossRef] [PubMed] [Google Scholar]
- RISC in Entamoeba histolytica: Identification of a Protein-Protein Interaction Network for the RNA Interference Pathway in a Deep-Branching Eukaryote. Johnson PJ, editor. mBio. 2021;125:e01540-21. Oct 26 [accessed 2023 Nov 9] Available from: https://journals.asm.org/doi/10.1128/mBio.01540-21
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Protein Phosphatase PP2C Identification in Entamoeba spp. BioMed Research International. 2021;2021 [accessed 2023 Nov 9] Available from: https://www.hindawi.com/journals/bmri/2021/5746629/
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Unraveling the relevance of the polyadenylation factor EhCFIm25 in Entamoeba histolytica through proteomic analysis. FEBS Open Bio. 2021;1110:2819-35. Sep 13 [accessed 2021 Oct 24] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8487052/
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- In silico analysis and molecular identification of an anaphase-promoting complex homologue from human pathogen Entamoeba histolytica. J Genet Eng Biotechnol. 2021;191:133. Dec [accessed 2023 Nov 9] Available from: https://jgeb.springeropen.com/articles/10.1186/s43141-021-00234-y
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Coordinated activity of amoebic formin and profilin are essential for phagocytosis. Molecular Microbiology. 2021;1163:974-95. Sep [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/10.1111/mmi.14787
- [CrossRef] [PubMed] [Google Scholar]
- Entamoeba histolytica trophozoites interact with the c-Met receptor at the surface of liver origin cells through the Gal/GalNAc amoebic lectin. Life[accessed 2023 Nov 9]. 2021;119:923. Available from: https://www.mdpi.com/2075-1729/11/9/923
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Bioinformatic analysis of two TOR Target of rapamycin-like proteins encoded by Entamoeba histolytica revealed structural similarities with functional homologs. Genes. 2021;128:1139. [accessed 2023 Nov 9] Available from: https://www.mdpi.com/2073-4425/12/8/1139
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- New insights on NETosis induced by entamoeba histolytica: Dependence on ROS from amoebas and extracellular MPO activity. Antioxidants. 2021;106:974. [accessed 2023 Nov 9] Available from: https://www.mdpi.com/2076-3921/10/6/974
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Protein SUMOylation is crucial for phagocytosis in Entamoeba histolytica trophozoites. International Journal of Molecular Sciences. 2021;2211:5709. [accessed 2023 Nov 9] Available from: https://www.mdpi.com/1422-0067/22/11/5709
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Two StAR-related lipid transfer proteins play specific roles in endocytosis, exocytosis, and motility in the parasitic protist Entamoeba histolytica. PLoS Pathogens. 2021;174:e1009551. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009551
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The atypical protein arginine methyltrasferase of Entamoeba histolytica EhPRMTA is involved in cell proliferation, heat shock response and in vitro virulence. Experimental Parasitology. 2021;222:108077. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/S001448942100014X
- [CrossRef] [PubMed] [Google Scholar]
- Endocytosis, signal transduction and proteolytic cleaving of human holotransferrin in Entamoeba histolytica. International Journal for Parasitology. 2020;5012:959-67. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/S0020751920302356
- [CrossRef] [PubMed] [Google Scholar]
- Uncovering the cyclic AMP signaling pathway of the protozoan parasite Entamoeba histolytica and understanding its role in phagocytosis. Front Cell Infect Microbiol. 2020;10:566726.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Characterization of Extracellular Vesicles from Entamoeba histolytica Identifies Roles in Intercellular Communication That Regulates Parasite Growth and Development. Saeij JPJ, editor. Infect Immun. 2020;8810:e00349-20. Sep 18 [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/IAI.00349-20
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Calcium modulates the domain flexibility and function of an a-actinin similar to the ancestral a-actinin. Proc Natl Acad Sci USA. 2020;11736:22101-12. Sep 8 [accessed 2023 Nov 13] Available from: https://pnas.org/doi/full/10.1073/pnas.1917269117
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A lysosomal hydrolase receptor, CPBF2, is associated with motility and invasion of the enteric protozoan parasite Entamoeba histolytica. Molecular and Biochemical Parasitology. 2020;239:111299. [accessed 2023 Nov 13] Available from: https://www.sciencedirect.com/science/article/pii/S0166685120300633
- [CrossRef] [PubMed] [Google Scholar]
- Ca2+-binding protein from Entamoeba histolytica EhCaBP6 is a novel GTPase. Biochemical and Biophysical Research Communications. 2020;5273:631-7. [accessed 2023 Nov 13] Available from: https://www.sciencedirect.com/science/article/pii/S0006291X20308810
- [CrossRef] [PubMed] [Google Scholar]
- Eh Rab21 associates with the Golgi apparatus in Entamoeba histolytica. Parasitology research. 2020;119:1629-40. [accessed 2023 Nov 13] Available from: https://idp.springer.com/authorize/casa?redirect_uri=https://link.springer.com/article/10.1007/s00436-020-06667-7&casa_token=S1MkdqctirsAAAAA:6zrti0T0EH7FUw341vwE1Z0S9kCRrQhhwxHGpzXHHSQUJ-sis2-pge181tmLsnFaN8S9Y6_TRhuHi2AESA
- [CrossRef] [PubMed] [Google Scholar]
- Two isotypes of phosphatidylinositol 3‐phosphate ‐ binding sorting nexins play distinct roles in trogocytosis in Entamoeba histolytica. Cellular Microbiology. 2020;223 Mar [accessed 2023 Nov 13] Available from: https://onlinelibrary.wiley.com/doi/10.1111/cmi.13144
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- EhC2B, a C2 domain-containing protein, promotes erythrophagocytosis in Entamoeba histolytica via actin nucleation. PLoS pathogens. 2020;165:e1008489. [accessed 2023 Nov 13] Available from: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1008489
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- EhRabB mobilises the EhCPADH complex through the actin cytoskeleton during phagocytosis of Entamoeba histolytica. Cellular microbiology. 2019;2110:e13071. [accessed 2023 Nov 9] Available from: https://onlinelibrary.wiley.com/doi/abs/10.1111/cmi.13071
- [CrossRef] [PubMed] [Google Scholar]
- HIF-1a modulates sex-specific Th17/Treg responses during hepatic amoebiasis. Journal of Hepatology. 2022;761:160-73. [accessed 2023 Nov 9] Available from: https://www.sciencedirect.com/science/article/pii/S0168827821020821
- [CrossRef] [PubMed] [Google Scholar]
- Plasma membrane damage repair is mediated by an acid sphingomyelinase in Entamoeba histolytica. PLoS Pathogens. 2019;158:e1008016. [accessed 2023 Nov 9] Available from: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1008016
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- EhVps23: A component of ESCRT-I that participates in vesicular trafficking and phagocytosis of Entamoeba histolytica. Frontiers in Cellular and Infection Microbiology. 2021;11:770759. [accessed 2023 Nov 13] Available from: https://www.frontiersin.org/articles/10.3389/fcimb.2021.770759/full
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A novel TLR4-binding domain of peroxiredoxin from Entamoeba histolytica triggers NLRP3 inflammasome activation in macrophages. Frontiers in Immunology. 2021;12:758451. [accessed 2023 Nov 9] Available from: https://www.frontiersin.org/articles/10.3389/fimmu.2021.758451/full
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The calcium binding protein EhCaBP6 is a microtubular-end binding protein in Entamoeba histolytica: EhCaBP6 is involved in chromosome segregation. Cell Microbiol. 2013;1512:2020-33. Dec [accessed 2023 Nov 13] Available from: https://onlinelibrary.wiley.com/doi/10.1111/cmi.12167
- [CrossRef] [PubMed] [Google Scholar]
- Entamoeba Chitinase is Required for Mature Round Cyst Formation. Alanio A, editor. Microbiol Spectr. 2021;91:e00511-21. Sep 3 [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/Spectrum.00511-21
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Stage-Specific De Novo Synthesis of Very-Long-Chain Dihydroceramides Confers Dormancy to Entamoeba Parasites. Blader IJ, editor. mSphere. 2021;62:e00174-21. Apr 28 [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/mSphere.00174-21
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Nuclear Factor Y NF-Y Modulates Encystation in Entamoeba via Stage-Specific Expression of the NF-YB and NF-YC Subunits. Johnson PJ, editor. mBio. 2019;103:e00737-19. Jun 25 [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/mBio.00737-19
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Role of inflammasomes in innate host defense against Entamoeba histolytica. Journal of Leucocyte Biology. 2020;1083:801-12. [accessed 2023 Nov 13] Available from: https://academic.oup.com/jleukbio/article-abstract/108/3/801/6884463
- [CrossRef] [PubMed] [Google Scholar]
- Androgens predispose males to monocyte-mediated immunopathology by inducing the expression of leukocyte recruitment factor CXCL1. Nature communications. 2020;111:3459. [accessed 2023 Nov 13] Available from: https://www.nature.com/articles/s41467-020-17260-y
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Trogocytosis by Entamoeba histolytica Mediates Acquisition and Display of Human Cell Membrane Proteins and Evasion of Lysis by Human Serum. Johnson PJ, editor. mBio. 2019;102:e00068-19. Apr 30 [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/mBio.00068-19
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The NF-kB Pathway: Modulation by Entamoeba histolytica and Other Protozoan Parasites. Front Cell Infect Microbiol. 2021;11:748404. Sep 14 [accessed 2021 Oct 22] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8476871/
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Entamoeba histolytica Interaction with Enteropathogenic Escherichia coli Increases Parasite Virulence and Inflammation in Amebiasis. Herbert DR, editor. Infect Immun. 2019;8712:e00279-19. Dec [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/IAI.00279-19
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Clinical features and gut microbiome of asymptomatic Entamoeba histolytica infection. Clinical Infectious Diseases. 2021;739:e3163-71. [accessed 2023 Nov 13] Available from: https://academic.oup.com/cid/article-abstract/73/9/e3163/5860457
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- South American Entamoeba dispar strains produce amoebic liver abscesses with different pathogenicities and evolutionary kinetics. Acta tropica. 2021;224:106114. [accessed 2023 Nov 13] Available from: https://www.sciencedirect.com/science/article/pii/S0001706X2100293X
- [CrossRef] [PubMed] [Google Scholar]
- The Entamoeba lysine and glutamic acid rich protein KERP1 virulence factor gene is present in the genomes of Entamoeba nuttalli, Entamoeba dispar and Entamoeba moshkovskii. Molecular and Biochemical Parasitology. 2020;238:111293. [accessed 2023 Nov 13] Available from: https://www.sciencedirect.com/science/article/pii/S0166685120300578
- [CrossRef] [PubMed] [Google Scholar]
- Pathogenic Entamoeba histolytica, but not Entamoeba dispar, induce neutrophil extracellular trap NET formation. Journal of Leukocyte Biology. 2019;1056:1167-81. [accessed 2023 Nov 13] Available from: https://academic.oup.com/jleukbio/article-abstract/105/6/1167/6935742
- [CrossRef] [PubMed] [Google Scholar]
- SARS-CoV-2 infection and parasitic diseases: A possible role for microbiome interaction? Parasitology International. 2021;85:102417. [accessed 2023 Nov 13] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8253655/
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Amoebic liver abscess in a COVID-19 patient: a case report. BMC Infect Dis. 2021;211:1134. Dec [accessed 2023 Nov 13] Available from: https://bmcinfectdis.biomedcentral.com/articles/10.1186/s12879-021-06819-9
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Functional Characterization of an Interferon Gamma Receptor-Like Protein on Entamoeba histolytica. Herbert DR, editor. Infect Immun. 2019;8711:e00540-19. Nov [accessed 2023 Nov 13] Available from: https://journals.asm.org/doi/10.1128/IAI.00540-19
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]