Translate this page into:
Unveiling the neuroprotective potential of chrysin on the pre-frontal cortex of adult male Wistar rats
*Corresponding author: Dr. Chiadikobi Lawrence Ozoemena, Department of Anatomy, College of Medicine, Enugu State University of Science and Technology, Parklane, Enugu, Nigeria. chiadikobi.ozoemena@esut.edu.ng
-
Received: ,
Accepted: ,
How to cite this article: Egwuatu IA, Ozoemena CL, Onuorah FK. Unveiling the neuroprotective potential of chrysin on the pre-frontal cortex of adult male Wistar rats. South Asian J Health Sci. 2024;1;62–8. doi: 10.25259/SAJHS_15_2023
Abstract
Objectives
Excessive free radicals in the human body predispose cells within the various systems to an imbalance and accumulation of oxygen-reactive species, known as oxidative stress. The central nervous system is not spared when it comes to these cell and tissue damages. Oxidative stress on the central nervous system may be responsible for anxiety, spatial memory impairment, neuronal cell depletion, and vacuole-tissue degeneration resulting from neurotoxicity. The use of chemotherapeutic agents such as doxorubicin has been implicated in the build-up of this imbalance between oxygen-reactive species and antioxidants. Therefore, it has become an area of research interest to seek antioxidant supplements that may offer neuroprotective effects. This study is aimed to evaluate the protective potential of chrysin on the pre-frontal cortex of male Wistar rats with doxorubicin-induced cognitive impairment.
Material and Methods
Thirty-five adult Wistar rats (180–200 g) were grouped into seven (1–7; n = 5). Group 1, the normal control, received normal saline treatment only throughout the study. Group 2 was administered with doxorubicin only for 21 days by intraperitoneal injection. Groups 3 and 4 were administered with chrysin in low and high doses for 21 days orally. Groups 5, 6 and 7 were exposed to doxorubicin and chrysin for 21 days intra-peritoneally and orally with low, medium and high doses, respectively.
Results
Anti-oxidative biomarkers analysed in Group 2 (doxorubicin-only) demonstrated a significant difference when compared to other groups. This corresponded to significant elevations in apoptotic indicators, inflammatory markers and histological lesions, which were indicative of cognitive impairment. 5, 7-dihydroxyflavone (chrysin) significantly mitigated and also reversed cognitive impairment caused by doxorubicin.
Conclusion
The data showed that chrysin protected against doxorubicin-induced cognitive impairment. This effect is probably made possible by suppressing oxidative stress, inflammation and apoptosis.
Keywords
Chrysin
Doxorubicin
Oxidative Stress
Cognitive impairment.
INTRODUCTION
Cognitive impairment refers to difficulty in remembering, learning, concentrating or making decisions that affect everyday life.[1] It can also be defined as frequent episodes of confusion or memory loss during the past 12 months.[1] It ranges from mild to severe. Severe conditions can cause one to lose the ability to speak or write, as well as the ability to comprehend the significance of anything, endangering independence.[1]
The pre-frontal cortex is a part of the brain located at the front of the frontal lobe, which is required to perform several functions, including orchestration of thoughts and actions following internal goals.[2] Organization, judgement, short-term memory, personality expression, behavioural and social control, and voice control are among the executive activities for which this brain area has been linked.[3, 4] Cognitive control is within the frontal cortex, impacting attention, impulse inhibition, prospective memory and cognitive flexibility.[4]
Doxorubicin (Adriamycin), is a cytotoxic anthracycline antibiotic typically used in combination with other chemotherapeutic agents in the management of numerous carcinomas, including breast cancer, Hodgkin’s and non-Hodgkin’s lymphoma, leukaemia, bronchogenic carcinoma, gastric, thyroid and prostate carcinoma.[5]
5, 7-dihydroxyflavone (chrysin) belongs to a class of chemicals called flavonoids, which are polyphenolic secondary metabolites found in plants and, thus commonly consumed by humans.[6] Chrysin occurs naturally in various plants and substances, such as honey, passionflower (Passiflora caerulea), silver linden and bee propolis (glue). Chrysin has high commercial and biological importance due to its multiple biological activities such as anticancer,[6] anti-angiogenesis,[7] antidiabetic,[8–10] anti-aging[11] and anti-allergic effects.[12]
The anterior region of the frontal lobes on both hemispheres contains the pre-frontal cortex and plays a critical role in regulating stress and is,thus affected directly by stress itself.[4] Thus, the idea that an accumulation of reactive oxygen species (ROS) within the brain jeopardises the pre-frontal cortex’s biochemical integrity through oxidative stress is enormously probable.[13] Chrysin is a potential protective agent in this experiment because it has anti-inflammatory, anti-oxidative, and anticancer effects.
This present study was designed to determine whether a 21-day chrysin treatment (2 mg/kg daily) protects the tissues of the pre-frontal cortex from doxorubicin-induced cognitive impairment in Wistar rats, as well as to ascertain the neuroprotective role of chrysin on oxidative stress and inflammation biomarkers of the pre-frontal cortex.
MATERIAL AND METHODS
Experimental animals
We acquired 35 albino Wistar male rats, each weighing between 150 and 200 grams, from the University of Nigeria’s Enugu campus and housed them in adequately ventilated cages. Before experimental manipulation, they were allowed two weeks to acclimate. Additionally, the rats had unrestricted access to conventional livestock feed and clean water. Using an electronic weighing scale, the animals’ weights were noted before, during, and after the study. The procedures of this study were conducted according to the Animal Care and Use Standard Operating Procedures and Guidelines (SOPGs), and ethical approval was obtained from the Ethics and Research Committee of the Faculty of Basic Medical Sciences, Enugu State University of Science and Technology with ethical right permission number: ESUCOM/ FBMS/ETR/2022/013.
Drugs
The phytochemical, chrysin (5, 7-dihydroxyflavone) was purchased from Sigma-Aldrich Company, 3050 Spruce Street, St. Louis, USA, and doxorubicin was purchased from Alpha pharmaceutical store in Enugu metropolis, Nigeria.
Experimental design
| Groups | Rats | Treatment | |
|---|---|---|---|
| 1 | Control | 5 | 0.2 mL of normal saline orally for 21 days. |
| 2 | DOX only | 5 | 2 mg/kg BW of doxorubicin given weekly for 21 days via Intra-peritoneal injection |
| 3 | CHR low dose only | 5 | 50 mg/kg BW of chrysin given daily for 21 days via oral route. |
| 4 | CHR high dose only | 5 | 150 mg/kg BW of chrysin given daily for 21 days via oral route. |
| 5 | DOX + CHR low dose | 5 | 2 mg/kg BW of doxorubicin given weekly for 21 days via Intra-peritoneal injection with 50 mg/kg BW of chrysin given daily for 21 days via oral route. |
| 6 | DOX + CHR medium dose | 5 | 2 mg/kg BW of doxorubicin given weekly for 21 days via Intra-peritoneal injection with 100 mg/kg BW of chrysin given daily for 21 days via oral route. |
| 7 | DOX + CHR high dose | 5 | 2 mg/kg BW of doxorubicin given weekly for 21 days via Intra-peritoneal injection with 150 mg/kg BW of chrysin given daily for 21 days via oral route. |
DOX: Doxorubicin, CHR: Chrysin, BW: Body Weight
Behavioural assessment
Novel object recognition
The NOR test was performed 24 hours after the last dose of chrysin was administered. On the first day, habituation was conducted for 30 minutes. The following day, two identical objects were indicated in the familiarisation route at two different arena corners. After spending three minutes exploring the items in the arena, the animals were taken out and left for 15 minutes in their cage. Subsequently, the arena and its contents were sanitised using 20% ethanol before a novel object (NO), and one of the familiar objects (FOs) were introduced. The animals had three minutes to investigate the items in the choice trail.[14] The video tracking software of EthoVision® XT version 12 (Noldus, Wageningen, and Netherlands) was used to capture the exploration time and mobility in both tests. During the habituation phase, the distance travelled, and the speed was monitored. The amount of time the rat spent pointing its nose at things that were 2 cm or less away was used to compute the exploration time.
Biochemical assays
Following the behavioural assessments, cervical dislocation was done under proper anaesthesia. The brains were taken out, divided into two halves, and left in 30% sucrose for three hours at 4°C. They were then frozen instantly in liquid nitrogen, embedded in an optimal cutting temperature (OCT) compound (Thermo Fisher Scientific, Karlsruhe, Germany), and kept at −80°C according to the methodology described by Spijker.[15] The homogenates were prepared in Tris-ethylenediaminetetraacetic acid (EDTA) (49.78 mM) buffer centrifuged for 40 min at 10.000 rpm at 4°C, and the supernatant was decanted and centrifuged at 45,000 rpm for 30 minutes before use for the enzyme assays.[16]
Superoxide dismutase (SOD) activity
The assessment for superoxide dismutase (SOD) activity was conducted using Marklund & Marklund’s technique.[17] 2.8 mL Tris-EDTA and 100 µL Pyrogallol (2 mM) and 50 µL tissue homogenate were taken in the cuvette and scanned for 3 min at 420 nm wavelength. The quantity of the enzyme that reduces the rate of pyrogallol auto-oxidation by 50% is known as one unit of SOD activity, and it was measured in units/mg protein/min.
Catalase (CAT) activity
CAT activity in the concentration of H2O2 (S1) was assayed spectrophotometrically by the method proposed by Aebi[18] which states that ‘Catalase exerts a dual function that includes the decomposition of H2O2 to give H2O and O2. We monitored S1 at 240 nm. In the ultraviolet range, H2O2 showed a continual increase in absorption with decreasing wavelength. The decomposition of H2O2 was followed directly by the decrease in absorbance at 240 nm (Ɛ240 = 0.00394 ± 0.0002 L mmol–1 mm–1). The difference in absorbance (ΔA240) per unit time is a measure of the catalase activity. The enzymatic activity was expressed as Units (U)/mg protein (1U decomposes 1 µmol H2O2/min at pH 7 at 25°C).
Malondialdehyde and glutathione activities
Malondialdehyde (MDA) is a low molecular weight end-product of lipid hydroperoxide decomposition and is the most often measured as a index of lipid peroxidation. According to Satyam,[19] an iMark microplate absorbance reader was used to detect the optical density of MDA spectrophotometrically at 540 nm wavelength according to the manufacturer’s instruction (NWK-MDA01). The expression for the enzymatic activity was represented as mM/mL. Glutathione activity was assayed spectrophotometrically by the method of Wendel,[20] analysing glutathione (GSH)/Nicotinamide-adenine dinucleotide phosphate (NADPH)/glutathione reductase (GR) system, by the dismutation of H2O2 at 340 nm. Through the capability of glutathione reductase, with NADPH, to regenerate reduced glutathione (GSH) from oxidised glutathione (GSSG), the decrease in NADPH was continuously measured spectrophotometrically while the GSH concentration in the enzymatic cycle remained essentially constant. The test measures the enzyme activity indirectly, and as a result of the breakdown of H2O2, reduced GSH is converted to oxidised glutathione (GSSG). The GR in the test media regenerates GSSG back to GSH at the expense of NADPH. The expression for the enzymatic activity was nmol NADPH/min/mg protein.
Caspase-3 activity, IL-1β and IL-6 assay
The brain tissue homogenate;s caspase activity was quantified, and cells that are either suspected of undergoing or have already undergone apoptosis were first lysed to extract their intracellular contents as described.[21] The activity of caspase-3 was analysed by spectrophotometric detection of chromophore p-nitroanilide (pNA) cleaved from the DEVD peptide sequence. The chromophore pNA is released when the peptide is broken down by caspase-3, and it has a wavelength of between 400 and 405 nm, making it suitable for spectrophotometric measurement.[22] The colour reaction is closely correlated with the amount of caspase enzymatic activity present in the cell lysate, and this allows for comparison across all the groups. DEVDase activity was detected using the APOPCYTO caspase-3 colorimetric assay kit (MBL Co.) according to the manufacturer’s directions. IL-6 (interleukin-6) and IL-I (interleukin-1) platinum enzyme-linked immunosorbent assay (ELISA) kits for rats were used to ascertain the level of inflammatory response following the manufacturer’s instructions (MBS764577). This kit was based on sandwich enzyme-linked immune-sorbent assay technology and the optical difference absorbance was detected at 450 nm, then the concentration of both IL-1β and IL-6 were calculated.
Histological procedure
The hippocampus was removed to expose the pre-frontal cortex which was dissected away from the olfactory bulb and the corpus callosum according to the methodology by Spijker[15] and subjected to histological studies. It was fixed in a 10% formalin solution in phosphate buffer and was processed using a paraffin wax embedding medium. Tissue blocks of the pre-frontal cortex were sectioned using an automatic microtome and sections were stained with routine H&E stain for histological examination.
Statistical analysis
All quantitative data were analysed using GraphPad version 8 and SPSS Version 23 (IBM Corp., Armonk, NY, USA) software, using one-way Analysis of Variance (ANOVA) followed by Tukey’s comparison test. Significance was set at P < 0.05. The results were demonstrated in tables and bar charts to show the mean and standard deviation.
RESULTS
Biochemical result
| Groups | Mean ± Standard Deviation | MDA | Glutathione | Catalase |
|---|---|---|---|---|
| 1 | 12.07 ± 0.36 F | 7.07 ± 0.67F | 26.67 ± 0.65F | 36.61 ± 0.90F |
| 2 | 8.92 ± 0.96* | 8.97 ± 0.18* | 22.66 ± 0.15* | 30.17 ± 1.90* |
| 3 | 11.94 ± 0.32 F | 7.43 ± 0.24F | 25.43 ± 0.41F | 34.42 ± 1.10F |
| 4 | 10.67 ± 0.21 | 8.44 ± 0.13* | 24.84 ± 0.75 | 32.93 ± 1.80 |
| 5 | 10.10 ± 0.48 F | 7.82 ± 0.13 | 25.19 ± 0.15F | 34.63 ± 0.60F |
| 6 | 11.46 ± 0.46 F | 8.08 ± 0.11 | 24.61 ± 0.94 | 33.91 ± 0.30 |
| 7 | 11.56 ± 0.91 F | 7.91 ± 0.29 | 24.61 ± 0.51 | 32.71 ± 0.80 |
Values were expressed as Mean ± SD; *P < 0.05 showed a significant difference in groups 2 and 4 compared to other groups; FP < 0.05 showed a significant difference compared to groups 1, 3, 4, 5, 6 and 7, compared with group 2, respectively.
MDA = Malondialdehyde.
| Groups | Mean ± Standard Deviation | IL-6 | Caspase-3 |
|---|---|---|---|
| 1 | 131.09 ± 9.27F | 204.35 ± 5.36F | 4.98 ± 0.30 |
| 2 | 182.58 ± 9.27* | 238.48 ± 5.96* | 6.90 ± 0.19* |
| 3 | 138.58 ± 6.62F | 215.31 ± 0.60F | 5.40 ± 0.32F |
| 4 | 157.30 ± 2.65 | 219.94 ± 3.58 | 5.52 ± 0.16F |
| 5 | 150.28 ± 8.61F | 222.89 ± 4.17 | 5.61 ± 0.23F |
| 6 | 148.88 ± 5.30F | 222.47 ± 9.53 | 6.07 ± 0.23* |
| 7 | 150.28 ± 3.31F | 213.20 ± 3.58F | 5.93 ± 0.16*F |
Values were expressed as Mean ± SD; *P < 0.05 showed a significant difference in groups 1, 3, 4, 5, 6 and 7 compared with the positive control (group 2); FP < 0.05 showed a significant difference in group 1, 3, 4, 5, 6 and 7 when compared with group 2, respectively.
IL-6= Interleukin 6.
Histomorphological result
![(a) A photomicrograph of the control group and the neuronal cells are interspersed within the neural tissue (black arrows) (b) cytoarchitecture with very mild signs of the onset of tissue traumatic encephalopathy (red arrows). (c)–(f) Several neuronal cells interspersed within the neural tissue and their cytoarchitecture appears normal (black arrows). [Hematoxylin & Eosin stain x200]](/content/162/2024/1/2/img/SAJHS-1-2-62-g1.png)
- (a) A photomicrograph of the control group and the neuronal cells are interspersed within the neural tissue (black arrows) (b) cytoarchitecture with very mild signs of the onset of tissue traumatic encephalopathy (red arrows). (c)–(f) Several neuronal cells interspersed within the neural tissue and their cytoarchitecture appears normal (black arrows). [Hematoxylin & Eosin stain x200]
Neurobehavioral results
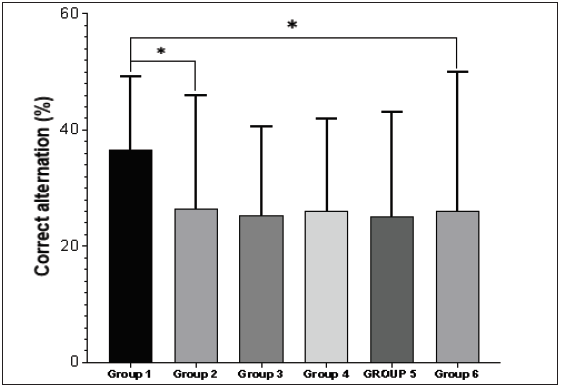
- Groups 1–6 demonstrate the percentage of correct alternation in the Y-maze test. There was a significant decrease in the percentage of correct alternation in group 2 when compared with group 1 (*P < 0.05). Also, group 7 possessed a significantly lower percentage of correct alternation relative to the control (*P < 0.05).
Bodyweight of experimental animals
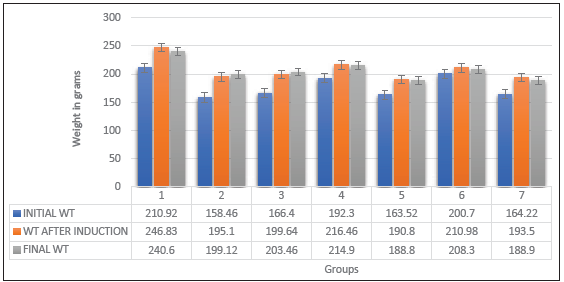
- Initial and final weight before and after induction. WT = Weight in grams.
DISCUSSION
Plant-based medicine is an essential part of healthcare, and one plant flavonoid in particular, chrysin, has a variety of pharmacological actions. Chrysin is used in the prevention of cancer and to treat erectile dysfunction, baldness, anxiety, inflammation, gout, HIV/AIDS and bodybuilding in humans.[10]
Memory impairments have been extensively described after chemotherapeutic protocols in experimental Wistar rats.[23,24] It is attributed to oxidative damage[23,25] and neuroinflammation.[26–29] Previous studies showed that with a dose of 2 mg/kg Body weight, doxorubicin induced cognitive impairments in adult Wistar rats over 21 days.[26] Another study also showed that a DOX dosage of 2 mg/kg BW induced toxicity in the pre-frontal cortex, hippocampus and hypothalamus.[27] This was the basis of the experimental design as demonstrated in Table 1.
Doxorubicin-induced cognitive impairment was studied by the estimation of antioxidant markers (MDA, GSH, SOD and CAT) [Table 2], anti-inflammatory markers (IL-1 and IL-6) and anti-apoptotic markers (caspase-3) see [Table 3] along with histopathological analysis of tissues of the pre-frontal cortex as demonstrated in Figure 1.
The histopathological examination demonstrated that in Figure 1a, several neuronal cells were interspersed within the neural tissue and there was normal cytoarchitecture. This group received phosphate buffer saline only. On the other hand, group two (positive control) received doxorubicin only and demonstrated mild onsets of tissue traumatic encephalopathy [Figure 1b]. Additionally, there was a loss of the round shape of normal neurons as well as the histological architecture, which was shown by the presence of darkish cytoplasm [Figure 1b]. This suggests that the pre-frontal cortex’s cytoarchitecture was cognitively hazardous when exposed to 2 mg/kg BW of DOX.[29] The pre-frontal cortex’s normal histological cytoarchitecture did not change in Group 3 (chrysin alone) [Figure 1c]. Groups 4, 5 and 6 likewise displayed no abnormalities in the pre-frontal cortex, which may indicate that chrysin treatment restored the normal neuronal characteristics [Figures 1d–1f].[29]
Thus, the administration of DOX could be said to have toxic effects on the tissues of the pre-frontal cortex, whereas the administration of chrysin brought about the restoration of these tissues to their normal conditions.[29]
The administration of DOX at a dose of 2 mg/kg/week for three consecutive weeks significantly triggered an oxidant effect as evidenced by reduced GSH and SOD levels, elevation of MDA levels, and reduction of catalase activity, when compared to the control group as demonstrated in Table 2. Also, the co-administration of varying doses of chrysin with DOX prominently restored the normal MDA and GSH levels as well as the CAT activity. Moreover, the chrysin-only treated group showed slight reductions in the SOD and GSH levels, catalase activity, and elevation of MDA levels compared to the control group.[29]
There was an increase in the mean levels of caspase-3 in the DOX group (group 2) when compared to group 1. This increase in the mean levels was the highest when compared to other groups. The protective groups (5, 6 and 7) for Caspase 3 showed an increase when compared to group 2, and so was dose-dependent, as there were increased mean levels of caspase-3 with higher doses of DOX. Apoptosis was involved in the pathogenesis of DOX-induced cognitive impairment and increased oxidative stress evoked by DOX triggered many signalling pathways, including the activation of caspase-3, leading to apoptosis [Table 3].[30–32]
In comparison to groups 3, 4, 5, 6 and 7, the DOX group’s mean level of IL1 and IL6 was lower than that of the control group [Table 3]. It has been demonstrated in earlier research that systemic DOX treatment raises pro-inflammatory cytokine levels, which encourage oxidative stress and cell death.[28] Early synaptic loss may also result via Caspase-3-driven apoptosis. According to the current study’s results, which showed a significant increase in IL-1 and IL-6 in the DOX group compared to controls, inflammation plays a major role in the pathophysiology of DOX-induced cognitive impairment. These findings are consistent with previous studies.[28] Uncertainty surrounds the main underlying mechanism driving this rise in inflammatory indicators, while potential triggers include reduced tissue antioxidant capacity, elevated ROS levels and consequent lipid peroxidation. It has been revealed recently that there is a correlation between elevated levels of oxidative stress and inflammatory mediators. Oxidative stress is believed to initiate inflammatory reactions by activating the NF-κB pathway, which in turn triggers the trans-activation of cytokines.[33] The current investigation showed that chrysin administration resulted in considerable drops in IL-1 and IL-6 levels, indicating a consistent protective effect of chrysin against DOX-mediated release of inflammatory mediators inside pre-frontal cortical tissues.
Behavioural studies were conducted to ascertain the effect of doxorubicin, with chrysin intervention [Figure 2]. Wistar rats were subjected to the Y maze test to test for cognitive impairment in the pre-frontal cortex. The outcome demonstrates a notable decline in the percentage of correct alternation in rats in group 2, which received DOX only when compared with group 1 (normal control). Group 6 had a significantly lower percentage of correct alternation relative to the control. Body weight was taken before and after induction, and the final weight was taken before sacrifice. Body weight was significantly increased in the control, DOX, and chrysin-only groups [Figure 3]. The body weight was reduced in protective groups 5, 6 and 7; the body weight did not significantly increase within the control group [Figure 3]. The adverse effect on body weight might be due to disruption of their metabolism due to their toxic effect, especially on neural tissues. These findings are in line with previous studies.[34,35]
In general, there was a significant decrease in the DOX-only groups compared to the negative control group. Also, the chrysin-treated groups showed a significant increase in body weight as compared to the DOX group. However, the mechanism by which chrysin brings about an increase in body weight was not determined.
CONCLUSION
From this research, it is shown that doxorubicin is an inducer of oxidative stress, in the pre-frontal cortex of adult male Wistar rats. Also, chrysin has a significant protective effect on the test subjects, which is dose-dependent. The search for a safer means of doxorubicin use in chemotherapy may, therefore, include co-administration or formulation with chrysin. Future studies should be focused on possible synergistic effects between doxorubicin and chrysin.
Ethical approval
The procedures of this study were conducted according to the Animal Care and Use Standard Operating Procedures and Guidelines (SOPGs), and ethical approval was obtained from the Ethics and Research Committee of the Faculty of Basic Medical Sciences, Enugu State University of Science and Technology with ethical right permission number: ESUCOM/ FBMS/ETR/2022/013.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
REFERENCES
- Cognitive impairment: A call for action, now. Atlanta, GA, USA: CDC; 2011. Feb
- The pre-frontal cortex: Categories, concepts, and cognition. Philos Trans R Soc Lond B Biol Sci. 2002;357:1123-36.
- [CrossRef] [PubMed] [Google Scholar]
- Pre-frontal structural and functional brain imaging findings in antisocial, violent, and psychopathic individuals: A meta-analysis. Psychiatry Res. 2009;174:81-8.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of the left pre-frontal cortex in language and memory. Proc Natl Acad Sci U S A. 1998;95:906-13.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- 8.24 - The Pulmonary Toxicity of Anticancer Agents. In: McQueen CA, ed. Comprehensive Toxicology (Second Edition). Oxford: Elsevier; 2010. p. :477-510.
- [Google Scholar]
- In vitro antitumor mechanisms of various Scutellaria extracts and constituent flavonoids. Planta Med. 2008;24:41-8.
- [CrossRef] [PubMed] [Google Scholar]
- Chrysin inhibits expression of hypoxia-inducible factor-1α through reducing hypoxia-inducible factor-1α stability and inhibiting its protein synthesis. Mol Cancer Ther. 2007;6:220-6.
- [CrossRef] [PubMed] [Google Scholar]
- A comparative study of flavonoid analogues on streptozotocin–nicotinamide induced diabetic rats: Quercetin as a potential antidiabetic agent acting via 11β-hydroxysteroid dehydrogenase type 1 inhibition. Eur J Med Chem. 2010;45:2606-12.
- [CrossRef] [PubMed] [Google Scholar]
- Inhibition of CCAAT/enhancer binding protein δ expression by chrysin in microglial cells results in anti‐inflammatory and neuroprotective effects. J Neurochem. 2010;115:526-36.
- [CrossRef] [PubMed] [Google Scholar]
- Chrysin protects mice from Staphylococcus aureus pneumonia. J Appl Microbiol. 2011;111:1551-8.
- [CrossRef] [PubMed] [Google Scholar]
- Protective role of chrysin against oxidative stress in d‐galactose‐induced aging in an experimental rat model. Geriatr Gerontol Int. 2012;12:741-50.
- [CrossRef] [PubMed] [Google Scholar]
- Chrysin attenuates allergic airway inflammation by modulating the transcription factors T-bet and GATA-3 in mice. Mol Med Rep. 2012;6:100-4.
- [CrossRef] [PubMed] [Google Scholar]
- The role of oxidative stress in the etiopathogenesis of chemotherapy-induced cognitive impairment (CICI)- “Chemobrain”. Aging Dis. 2016;7:307.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- In vivo neuroimaging and behavioural correlates in a rat model of chemotherapy-induced cognitive dysfunction. Brain Imaging Behav. 2018;12:87-95.
- [CrossRef] [PubMed] [Google Scholar]
- Quercetin down-regulates signal transduction in human breast carcinoma cells. Biochem Biophys Res Commun. 1995;208:425-31.
- [CrossRef] [PubMed] [Google Scholar]
- Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974;47:469-74.
- [CrossRef] [PubMed] [Google Scholar]
- Grape seed extract and zinc-containing nutritional food supplement prevents the onset and progression of age-related cataract in Wistar rats. J Nutr Health Aging. 2014;18:524-30.
- [CrossRef] [PubMed] [Google Scholar]
- Glutathione peroxidase. In: Methods Enzymol. Vol 77. Academic Press; 1981. p. :325-33.
- [CrossRef] [PubMed] [Google Scholar]
- Caspase functions in cell death and disease. Cold Spring Harbor perspectives in biology. 2013;5:a008656.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- [cited 2023 Dec 2]. Available from: https://www.creative-bioarray.com/support/caspase-activity-assay.htm
- Cognitive dysfunction induced by chronic administration of common cancer chemotherapeutics in rats. Metab Brain Dis. 2008;23:325-33.
- [CrossRef] [PubMed] [Google Scholar]
- Female rats induced with mammary cancer as a relevant animal model for doxorubicin‐induced chemobrain in vivo. Clin Exp Pharmacol Physiol. 2016;43:862-3.
- [CrossRef] [PubMed] [Google Scholar]
- Superoxide induces protein oxidation in plasma and TNF-α elevation in macrophage culture: Insights into mechanisms of neurotoxicity following doxorubicin chemotherapy. Cancer Lett. 2015;367:157-61.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Chemobrain: A critical review and causal hypothesis of the link between cytokines and epigenetic reprogramming associated with chemotherapy. Cytokine. 2015;72:86-96.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Doxorubicin-induced systemic inflammation is driven by upregulation of toll-like receptor TLR4 and endotoxin leakage. Cancer Res. 2016;76:6631-42.
- [CrossRef] [PubMed] [Google Scholar]
- Chemotherapy-induced cognitive impairment is associated with cytokine dysregulation and disruptions in neuroplasticity. Mol Neurobiol. 2019;56:2234-43.
- [CrossRef] [PubMed] [Google Scholar]
- Caffeic acid phenethyl ester counteracts doxorubicin-induced chemobrain in Sprague-Dawley rats: Emphasis on the modulation of oxidative stress and neuroinflammation. Neuropharmacology. 2020;181:108334.
- [CrossRef] [PubMed] [Google Scholar]
- Nose-to-brain delivery of chrysin transpersonal and composite vesicles in doxorubicin-induced cognitive impairment in rats: Insights on formulation, oxidative stress, and TLR4/NF-kB/NLRP3 pathways. Neuropharmacology. 2021;197:108738.
- [CrossRef] [PubMed] [Google Scholar]
- Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. J Mol Cell Cardiol. 2012;52:1213-25.
- [CrossRef] [PubMed] [Google Scholar]
- Self-assembled betulinic acid protects doxorubicin-induced apoptosis followed by reduction of ROS-TNF-alpha-caspase-3 activity. Biomed Pharmacother. 2015;72:144-157.
- [CrossRef] [PubMed] [Google Scholar]
- Vitexin attenuates acute doxorubicin cardio-toxicity in rats via the suppression of oxidative stress, inflammation, and apoptosis and the activation of FOXO3a. Exp Ther Med. 2016;12:1879-84.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Protective role of vitamin E and catechin on doxorubicin-induced cardiotoxicity in rats. Cancer Research Therapy and Control. 2001;11:175-182.
- [Google Scholar]
- Prevention of doxorubicin toxicity by catecin. Cancer Lett. 1995;98:126.
- [CrossRef] [PubMed] [Google Scholar]