Translate this page into:
Prevalence and risk factors for chronic kidney disease of unknown aetiology (CKDu) among industrial workers
* Corresponding author: Ravibabu Kalahasthi, Department of Biochemistry, Regional Occupational Health Centre Southern, Kannamangala post Poojanahalli Devanahalli, Bangalore, India. kalahasthi20012002@yahoo.co.in
-
Received: ,
Accepted: ,
How to cite this article: Kalahasthi R, Jakkam S, Jamalpur RP, Adepu VK, Nagaraju R. Prevalence and risk factors for chronic kidney disease of unknown aetiology (CKDu) among industrial workers. South Asian J Health Sci. 2024;1:77–82. doi: 10.25259/SAJHS_2_2024
Abstract
Objectives
Industrial workers are facing an occurrence of high risk factors. Exploring the frequency of CKDu and associated risk factors in industrial workers is needed. This study examined the CKDu prevalence and associated risk factors in industrial workers. Design: Cross section study.
Material and Methods
We enrolled 134 (83 male and 41 female) industrial workers. Modifying diet in renal disease (MDRD) equation was used to determine the estimated glomerular filtration rate (eGFR). The CKDu stages 1–5 were assessed using Kidney Disease: Improving Global Outcomes (KIDGO) guidelines.
Results
The mean eGFR was reported to be 99.2 for male workers and 85.2 ml/min per 1.73 m2 for female workers. The average eGFR among male workers was higher than female workers. 43.3% of workers had normal eGFR (> 90), while 52.2% of workers had a mild reduction (89–60), and 4.5% of workers had a moderate loss (59–45). No cases of severe loss and kidney failure (< 15) were noted. CKDu stage 2 was significantly higher in female workers than male workers. The increased odds ratio (OR) of CKDu stage 3 (< 60) was noted among male workers, with an increase in age, overweight, obesity, hypertension, diabetes, habits of smoking, and alcohol consumption.
Conclusion
The present study noted a lower eGFR and a higher OR of CKDu in industrial workers with risk factors. To avoid the progression from moderate severe loss and kidney failure, suggested weight reduction, avoiding smoking and alcohol consumption, and controlling diabetes and hypertension.
Keywords
Industrial workers: CKDu
Risk factors
eGFR
MDRD
KIDGO
INTRODUCTION
Chronic Kidney Disease of unknown aetiology (CKDu) is a non-communicable disease, and an advanced stage of CKD requires a high cost of treatment. Several epidemiological studies have reported an occurrence of CKDu (< 60 ml/min per 1.73 m2) in the range of 1 to 11%. The CKDu mortality rate was reported at 11.1 per 1 00,000 as per the records in 2010.[1] The endemics of kidney diseases also termed chronic kidney disease of nontraditional cause (CKDnT), and Mesoamerican endemic nephropathy (MEN). Reduced estimated glomerular filtration rate (eGFR) values were seen in farmers, construction workers, and sugar cane cutters at 16%, 9%, and 2%, respectively.[2] CKDu risk was associated with factors such as extreme heat, dehydration, and the use of agrochemicals in the agriculture community.[3] Lebov et al. (2015) found a significant relationship between pesticide exposure and end-stage renal disease among wives of pesticide applicators.[4] The manifestation of CKDu was observed in several countries: El Salvador,[5] Nicaragua,[6] Costa Rica,[7] Sri Lanka,[8] Egypt,[9] and India.[10] Most of these studies indicated that the use of agrochemicals, heat stress, heavy metal exposure, genetic predisposition, and the use of alcohol and tobacco products are related to the risk of CKDu. Recent studies on industrial workers have observed a high prevalence of Cardiovascular disease (CVD),[11] overweight,[12] hypertension[13] and occupational stress.[14]
These risk factors may influence the progression of CKDu stages. CKDu prevalence and associated risk factors in industrial workers need to be addressed. In this study, we aimed to investigate CKDu prevalence and associated risk factors in industrial workers.
MATERIAL AND METHODS
This study design is cross-sectional. The CKDu stages and associated risk factors were assessed among 134 industrial workers (Male=93 and female=41) engaged in industrial establishments in Karnataka state. These workers were involved in bearing and flavours manufacturing units, having five years of average experience. The convenience sampling method was used. Before participating in the study, each worker gave written, informed consent. The Institutional Ethics Committee (IEC) approved the study protocol in accordance with the National Ethical Guidelines for Biomedical and Health Research in India. Workers above 18 years of age and willing to take part were included. The workers not willing and having major health problems with medication were excluded.
The data on gender, age, habits of smoking, and alcohol consumption were collected using a questionnaire. The workers’ height was measured in centimetres using tape and their weight in kilograms with scales, while they wore lightweight clothes without shoes. The subject weight (kg) and height (metre squared) were used to evaluate Body mass index (BMI) and expressed as kg/m2. The Asia-Pacific BMI classification system was used to classify the subjects: underweight (≤18.5), normal weight (>18.5 to 22.9), overweight (23 to 24.9), and obese (>25) kg/m2.[15] The waist circumference (WC) measured the bottom of the ribs and the top of the hips of the study subjects by using tape. The categories of WC risk present or absent are done using Asian Indian action levels.[16]
The categories of gender, age, habits of smoking, alcohol consumption, and waist circumference are included as risk factors. The smoking status is defined as either non-smoker or subjects who smoke more than one cigarette per day. The definition of alcohol consumption status is considered as never drinking and currently drinking.
Blood pressure was measured using the OMRON HEM-7211E model. The subjects’ right arm, seated position, and three measurements were taken with a 5-minute rest between the measurements. The presence of hypertension was classified using the Joint National Committee (JNC) 8th report.[17] The worker is classified as hypertensive if their systolic blood pressure (SBP) is 140 mmHg or diastolic blood pressure (DBP) is 90 mmHg or above.
Blood collection: One millilitre of whole blood was drawn from each employee and left to stand at room temperature for half an hour. Serum samples were separated by centrifuging at 4000 rpm for 10 minutes at 4°C and used for glucose and creatinine estimation. A modified kinetic Jaffe reaction measures the level of serum creatinine.[18] The glucose-oxidase method was used to estimate glucose concentration. An American Diabetic Association criterion was used to evaluate diabetes in workers. If the worker has fasting serum glucose > 100 mg/dL, consider them as diabetic.
Measurement of eGFR: It was calculated using a modified Modification of Diet in Renal Disease (MDRD) equation.[19] The eGFR was expressed in mL/min/1.73 m2 of body surface area. The CKDu stages were assessed using Kidney Disease: Improving Global Outcomes (KIDGO) guidelines. The CKDu stages were categorised into stage one normal (≥ 90), stage two mild (89-60), stage three moderate (59-30), stage four –severe (29-15), and stage five-Kidney failure (< 15).
Statistical Analysis: The data analysis was done using Statistical Package for the Social Sciences (SPSS) version 23 software (IBM Corp., Armonk, NY, USA). The demographic details of workers were presented as numbers and percentages. The eGFR values as mean ± SD and eGFR categories were presented as proportions. The Odds ratio (OR) risk CKDu stage 3 was assessed using an unadjusted Odds ratio with a 95% confidence interval. The level of significance was considered P< 0.05.
RESULTS
Table 1 describes the industrial workers’ demographics. Male workers were found to be in a higher proportion than female workers. The age category of 26–33 years was high compared to other age groups. BMI was categorised as per the Asian population classification. The BMI of workers indicated that 53% were in the normal range, 28.4% were overweight, and 8.2% were obese. Only 10.4% of workers were underweight. Alcohol consumption and smoking habits were recorded at 25.4% and 11.9%, respectively. Hypertension and diabetes were noted in 15.7% and 17.2%, respectively.
| Variables | n (%) | eGFR Mean ± SD | eGFR categories | ||
|---|---|---|---|---|---|
| < 60 | > 60–< 90 | > 90 | |||
| Gender | |||||
| Female | 41 (30.6) | 85.2 ± 29.4 | 01 (2.4) | 31 (75.6) | 09 (22.0) |
| Male | 93 (69.4) | 99.2 ± 45.7 | 05 (5.4) | 39 (41.9) | 49 (52.7) |
| Age (years) | |||||
| 18-25 | 27 (20.1) | 101.7 ± 59.4 | 01 (3.7) | 10 (37.0) | 16 (59.3) |
| 26-33 | 48 (35.8) | 91.2 ± 33.5 | 02 (4.2) | 27 (56.3) | 19 (39.6) |
| 34-41 | 34 (25.4) | 99.9 ± 42.6 | 00 (0.0) | 20 (58.8) | 14 (41.2) |
| > 41 | 25 (18.7) | 88.9 ± 31.3 | 03 (12.0) | 13 (52.0) | 09 (36.0) |
| BMI (Kg/m2) | |||||
| < 18.5 (underweight) | 14 (10.4) | 85.2 ± 14.9 | 00 (0.0) | 08 (57.2) | 06 (42.8) |
| 18.5-22.9 (normal) | 71 (53.0) | 99.4 ± 51.9 | 03 (4.2) | 37 (52.1) | 31 (43.7) |
| 23-27.5 (overweight) | 38 (28.4) | 91.3 ± 28.7 | 02 (5.3) | 19 (50.0) | 17 (44.7) |
| >28 (obese) | 11 (8.2) | 90.6 ± 25.2 | 01 (9.1) | 6 (54.5) | 4 (36.4) |
| Hypertension | |||||
| Yes (SBP≥140/ DBP≥90 mm Hg) | 21 (15.7) | 92.2 ± 38.8 | 02 (9.5) | 08 (38.1) | 11 (52.4) |
| No | 113 (84.3) | 109.5 ± 53.9 | 04 (3.5) | 62 (55.8) | 47 (41.6) |
| Diabetes | |||||
| Yes (FBS >100 mg/dl) | 23 (17.2) | 90.4 ± 37.4 | 02 (8.7) | 10 (43.5) | 11 (47.8) |
| No | 111 (82.8) | 95.9 ± 47.7 | 04 (3.6) | 60 (54.1) | 47 (42.3) |
| Waist circumference(risk) | |||||
| Yes (M ≥ 90 cm & F ≥ 80 cm) | 60 (44.8) | 95.4 ± 47.7 | 02 (3.3) | 32 (53.3) | 26 (43.3) |
| No | 74 (55.2) | 95.0 ± 37.0 | 04 (5.4) | 38 (51.4) | 32 (43.2) |
| Smoking | |||||
| Yes | 16 (11.9) | 98.7 ± 41.0 | 01 (6.3) | 07 (43.7) | 08 (50.0) |
| No | 118 (88.1) | 94.4 ± 42.0 | 05 (4.2) | 63 (53.4) | 50 (42.4) |
| Alcohol consumption | |||||
| Yes | 34 (25.4) | 97.0 ± 45.0 | 03 (8.8) | 16 (47.1) | 15 (44.1) |
| No | 100 (74.6) | 942 ± 40.8 | 03 (3.0) | 54 (54.0) | 43 (43.0) |
eGFR: estimated glomerular filtration rate, SD: Standard deviation, BMI: Body Mass Index, SBP: Systolic blood pressure, DBP: Diastolic blood pressure, FBS: Fasting blood sugar
Table 2 shows the Odds ratio (OR) CKDu (stage 3) among industrial workers. The OR risk CKDu stage 3 was assessed using an unadjusted Odds ratio with a 95% confidence interval. The OR was increased in males, workers increased in age, obese and overweight, and the presence of hypertension, diabetes, smoking, and alcohol consumption.
| Variables | eGFR | Unadjusted OR (95% CI) | P value | |
|---|---|---|---|---|
| Absent (> 60) n (%) | Present (< 60) n (%) | |||
| Gender | ||||
| Female | 40 (97.6) | 01 (2.4) | Reference | |
| Male | 88 (94.6) | 05 (5.4) | 2.27 (0.257 – 20.092) | 0.460 |
| Age (years) | ||||
| 18–25 | 26 (96.3) | 01 (3.7) | Reference | |
| 26–33 | 46 (95.8) | 02 (4.2) | 1.130 (0.097 – 13.076) | 0.921 |
| 34–41 | 34 (100) | 00 (0.0) | 0.256 (0.010 – 6.540) | 0.409 |
| >41 | 22 (88.0) | 03 (12.0) | 3.545 (0.343 – 36.562) | 0.287 |
| BMI (Kg/m2) | ||||
| < 18.5 (underweight) | 14 (100) | 00 (0.0) | 0.674 (0.033 – 13.78) | 0.798 |
| 18.5–22.9 (Normal) | 68 (95.8) | 03 (4.2) | Reference | |
| 23–27.5 (overweight) | 36 (94.7) | 02 (5.3) | 1.259 (0.201 – 7.88) | 0.805 |
| >28 (obese) | 10 (90.9) | 01 (9.1) | 2.266 (0.214 – 23.97) | 0.496 |
| Hypertension | ||||
| No | 109 (96.5) | 04 (3.5) | Reference | |
| yes | 19 (90.5) | 02 (9.5) | 2.547 (0.438 – 14.818) | 0.297 |
| Diabetes | ||||
| No | 107 (96.4) | 04 (3.6) | Reference | 0.242 |
| Yes | 21 (91.3) | 02 (8.7) | 2.868 (0.490 – 16.772) | |
| Waist circumference (risk) | ||||
| No | 70 (94.6) | 04 (5.4) | Reference | 0.318 |
| Yes | 58 (96.7) | 02 (3.3) | 2.413 (0.426 – 13.652) | |
| Smoking | ||||
| No | 113 (95.8) | 05 (4.2) | Reference | 0.716 |
| Yes | 15 (93.7) | 01 (6.3) | 1.506 (0.164 – 13.783) | |
| Alcohol consumption | ||||
| No | 97 (97.0) | 03 (3.0) | Reference | 0.175 |
| Yes | 31 (91.2) | 03 (8.8) | 3.129 (0.600 – 16.303) | |
eGFR: estimated glomerular filtration rate, OR: Odds ratio, BMI: Body Mass Index, CI: Confidence Interval
Figure 1 shows the prevalence of different CKDu stages (1 to 5) among industrial workers: 43.3%, 52.2%, and 4.5% of workers, respectively, were in stages 1, 2, and 3. Stage 4 (severe loss) and 5 (kidney failure) were not found.
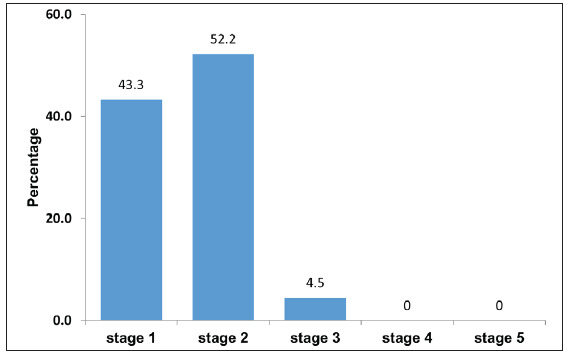
- Prevalence of Chronic Kidney Disease of unknown aetiology (CKDu) stages among industrial workers.
The prevalence of CKDu stages in male and female industrial workers was reported in Figure 2. The CKDu stage-2 was significantly increased in female workers compared to male workers. In male workers, CKDu stage 3 was higher than in female workers, while the difference was not statistically significant.
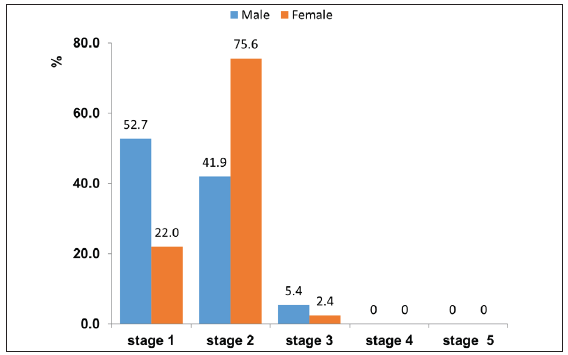
- Prevalence of Chronic Kidney Disease of unknown aetiology (CKDu) stages in male and female industrial workers.
DISCUSSION
In this study, we assessed the average prevalence of eGFR (<60 ml/minute/1.73m2) in industrial workers. In our current study, we noted that the average eGFR was 94.9, and the prevalence of eGFR was 4.5% among industrial workers. O’Callaghan-Gordo et al. reported a mean eGFR of 105 and a prevalence of eGFR of 1.6% in a population-based study.[20] Bragg- Gresham et al. (2020) reported that CKDu prevalence was 2.0% in the population living in Punjab state in north India.[21] In the current study, we found a reduced mean eGFR and a high prevalence of eGFR in industrial workers compared to population-based findings. Tatapudi et al reported 13% of CKDu prevalence in the population residing in the endemic area of Uddanam in Andhra Pradesh.[22] In the present study, we reported 4.5% among industrial workers, which was lower than the endemic area of Andhra Pradesh.
In the current study, we noted 43.3%, 52.2%, and 4.5% of workers found in stages 1, 2, and 3, respectively. No cases were found in CKDu stages 4 (severe loss) and 5 (renal failure). According to Soni et al., populations with an average age of 57 years had 18.6% in stage 1, 64.7% in stage 2, and 16.7% in stage 3. There were also 22.3% cases of hypertension and 25.6% cases of diabetes.[23] In the current study, we reported that stage 3 is 4.5%, with an average of 33.5 years, 9.5% hypertension, and 8.7% diabetics. Another survey from Karnataka reported 6.3% of CKDu prevalence in rural populations with a high prevalence of hypertension.[24]
During the study, we investigated the prevalence of CKDu and the occurrence of risk factors such as age, BMI, hypertension, diabetes, and habits of smoking and alcohol consumption. We observed that CKDu stage 2 was considerably elevated among female workers than male workers. Senevirthan et al. also reported similar findings concerning CKDu stage 2.[25] Regarding the prevalence of CKDu stage 3, we reported a higher proportion of male workers. Similar findings were observed in population-based studies in India.[26,27] An increase in age was noted to decrease eGFR values and increase the prevalence of CKDu in population-based studies.[28,29] This study reported similar findings with the increase in age, reduced eGFR, and higher prevalence of CKDu.
During the study, we noted a higher OR of CKDu in overweight and obese workers compared to a normal BMI. Herrington and colleagues noted that obesity or overweight status was linked to an increased chance of developing chronic kidney disease.[30] The current study reported an increased OR for CKDu prevalence in WC. He et al. also observed similar findings with respect (WC) risk with CKD in Chinese adults.[31] In the present study, we noted a higher odds ratio of CKDu in workers with hypertension as compared to normal. Hypertension is a significant risk factor for renal disease prognosis, and the high prevalence of hypertension is evidenced by the high prevalence of CKDu.[32] Rainer noted that diabetes, hypertension, and smoking are risk factors in the progression of CKD.[33] In this study, we compared the prevalence of CKDu with diabetes and found low eGFR values and a high prevalence of CKDu. Yacoub et al. noted that cigarette smoking causes a higher risk for CKD.[34] We also noted the higher OR of CKDu among workers who have the habit of smoking. Lee et al. found that subjects with higher alcohol intake had low levels of eGFR as compared to non-drinkers.[35] This study also noted similar findings, such as a low eGFR and a high CKDu.
LIMITATION
The present study was conducted on a small sample size. In future studies, we recommend a large number of samples with the incorporation of risk factors for CKDu.
CONCLUSION
The present study noted a lower eGFR and a higher odds ratio of CKDu in industrial workers with risk factors such as age, overweight, obesity, WC risk, hypertension, diabetes, smoking, and alcohol consumption. To avoid the progression from moderate to Severe loss and kidney failure, we suggested weight reduction, avoiding smoking and alcohol consumption, and controlling diabetes and hypertension.
Acknowledgements
The authors are thankful to the Director, ICMR-National Institute of Occupational Health, for his support and encouragement to conduct this study. The authors are also thankful to Mrs. Thara, Senior Technical Officer, and Mr. Rajeev Yadav, Technician C, for their assistance in the collection of data and samples.
Ethical approval
The research/study was approved by the Institutional Review Board at Regional Occupational Health Centre (Southern), number 2, dated 13-12-2018.
Declaration of patient consent
The authors certify that they have obtained all appropriate participants consent.
Financial support and sponsorship
ICMR –National Institute of Occupational Health Meghani Nagar, Ahmedabad-380016, Gujarat, India.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript, and no images were manipulated using AI.
References
- Chronic kidney disease of unknown etiology in agricultural communities. MEDICC review. 2014;16:9-15.
- [CrossRef] [PubMed] [Google Scholar]
- Heat stress, hydration and uric acid: A cross-sectional study in workers of three occupations in a hotspot of Mesoamerican nephropathy in Nicaragua. BMJ open. 2016;6:e011034.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Chronic kidney disease in Nicaraguan sugarcane workers: A historical, medical, environmental, and ethical analysis. Internet J Third World Med. 2016;12:1-16.
- [Google Scholar]
- Pesticide exposure and end-stage renal disease risk among wives of pesticide applicators in the Agricultural Health Study. Environ. Res. 2012;143:198-210.
- [Google Scholar]
- Epidemiology of chronic kidney disease in adults of Salvadoran agricultural communities. MEDICC Rev. 2011;16:23-30.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of and risk factors for chronic kidney disease in rural Nicaragua. Nephrology Dialysis Transplantation. 2011;26:2798-805.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Chronic kidney disease in Costa Rica. Kidney Int. 2005;68:S31-33.
- [CrossRef] [PubMed] [Google Scholar]
- Environmental contamination and its association with chronic kidney disease of unknown etiology in the North Central region of Sri Lanka. New Delhi. Centre for Science and Environment. 2012;37:1-37. Available from: http://www.cseindia.org/userfi les/sri_lanka_fi nal_report.pdf
- [Google Scholar]
- End-stage renal disease in the El-Minia Governorate, Upper Egypt: An epidemiological study. Saudi J Kidney Dis Transpl. 2011;22:1048-54.
- [PubMed] [Google Scholar]
- What do we know about chronic kidney disease in India: First report of the Indian CKD Registry. BMC Nephrology. 2012;13:1-8.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Cardiovascular risk factors among industrial workers: a cross–sectional study from eastern Nepal. J Occup Med Toxicol. 2016;11:1-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Prevalence and risk for overweight among employees in a leading auto-mobile industry in India. International Journal of Community Medicine and Public Health. 2021;8:5318-28.
- [Google Scholar]
- The Correlation between work stress and hypertension among industrial workers: A Cross-sectional study. IOP Conference Series. Earth and Environmental Science; Bristol. 2020;441:1.
- [Google Scholar]
- Occupational stress and associated risk factors among 13,867 industrial workers in China. Frontiers in public health. 2022;10:945902.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Comparison of World Health Organization and Asia-Pacific body mass index classifications in COPD patients. Int J Chron Obstruct Pulmon Dis. 2017;12:2465-75.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Waist circumference cut-off points and action levels for Asian Indians for identification of abdominal obesity. Int J Obes (Lond). 2006;30:106-11.
- [CrossRef] [PubMed] [Google Scholar]
- Evidence-based guideline for the management of high blood pressure in adults: Report from the panel members appointed to the Eight Joint National Committee (JNC 8) JAMA. 2014;311:507-20.
- [CrossRef] [PubMed] [Google Scholar]
- Creatinine Estimation and Interference. Indian J Clin Biochem. 2013;28:210-1.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-12.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Prevalence of and risk factors for chronic kidney disease of unknown aetiology in India: Secondary data analysis of three population-based cross-sectional studies. BMJ Open. 2019;9:e023353.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Population-based comparison of chronic kidney disease prevalence and risk factors among adults living in the Punjab, Northern India and the USA (2013-2015) BMJ Open. 2020;10:e040444.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- High prevalence of CKD of unknown etiology in Uddanam, India. Kidney Int Rep. 2019;4:380-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Prevalence of chronic kidney disease among severely gas-exposed survivors in Bhopal, India. Natl Med J India. 2023;36:5-10.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of chronic kidney disease in India - Where are we heading? Indian J Nephrol. 2015;25:133-5.
- [PubMed] [PubMed Central] [Google Scholar]
- Risk factors associated with disease progression and mortality in chronic kidney disease of uncertain etiology: A cohort study in Medawachchiya, Sri Lanka. Environ Health Prev Med. 2012;17:191-8.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Chronic kidney disease of unknown etiology in India: What do we know and where we need to go. Kidney Int Rep. 2021;6:2743-51.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Chronic kidney disease of unknown etiology: Case definition for India - A Perspective. Indian J Nephrol. 2020;30:236-40.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Age- and gender-specific reference values of estimated GFR in Caucasians: the Nijmegen Biomedical Study. Kidney Int. 2007;72:632-7.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic kidney disease of unknown etiology in a tertiary care teaching hospital. Cureus. 2023;15:e35446.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Body-mass index and risk of advanced chronic kidney disease: Prospective analyses from a primary care cohort of 1.4 million adults in England. PLoS One. 2017;12:e0173515.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The association of chronic kidney disease and waist circumference and waist-to-height ratio in Chinese urban adults. Medicine (Baltimore). 2016;95:e3769.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Hypertension as cardiovascular risk factor in chronic kidney disease. Circ Res. 2023;132:1050-63.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors in the progression of chronic kidney disease. European Endocrinology (2):41-6.
- [Google Scholar]
- Association between smoking and chronic kidney disease: A case control study. BMC Public Health. 2010;10:731.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Effect of alcohol consumption on kidney function: population-based cohort study. Sci Rep. 2021;11:2381.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]